Журнал «Медицина неотложных состояний» Том 22, №2, 2026
Вернуться к номеру
Нанотехнології проти гострого панкреатиту: інноваційні стратегії та перспективи (систематичний огляд)
Авторы: Чуклін С.М., Чуклін С.С.
Медичний центр Святої Параскеви, м. Львів, Україна
Рубрики: Медицина неотложных состояний
Разделы: Справочник специалиста
Версия для печати
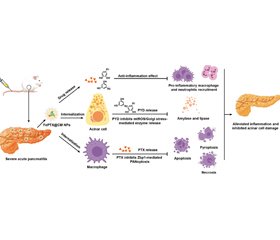
Актуальність. Гострий панкреатит (ГП) — гостре запалення підшлункової залози, для якого на сьогодні не існує специфічного лікування. Наявні підходи (протизапальні, антиоксидантні) малоефективні, зокрема через низьку біодоступність і коротку дію препаратів. Це зумовлює потребу в інноваційних методах терапії з адресною доставкою ліків до ураженої залози та мінімізацією побічних ефектів. Нанотехнології постають як перспективна платформа такої таргетної терапії: наноматеріали можуть прицільно доставляти препарати у вогнище запалення та знижувати їх системну токсичність. Метою огляду є аналіз сучасних досліджень застосування нанотехнологій у терапії ГП. Матеріали та методи. Проведено систематичний пошук літератури (PubMed, Scopus, Google Scholar) за настановами PRISMA 2020. Серед 316 знайдених джерел критеріям включення відповідали 62 дослідження (переважно доклінічні на тваринах) з нанотерапії ГП. Результати. Досліджено широкий спектр нанотерапевтичних підходів при ГП, включно з різними типами наноносіїв (полімерні, ліпідні, вуглецеві, органометалеві, біоміметичні). Нанопрепарати забезпечують адресну доставку ліків, контрольоване вивільнення, підвищення біодоступності та зниження системної токсичності. У моделях ГП нанотерапія продемонструвала потужний протизапальний і антиоксидантний ефект: зменшувала рівні цитокінів, нейтралізувала надлишкові вільні радикали, стабілізувала функцію мітохондрій та захищала ацинарні клітини від ушкодження. Загалом наностратегії поліпшували перебіг ГП в експериментальних моделях і перевершували за ефективністю традиційну терапію. Висновки. Нанотерапія є перспективним підходом до лікування гострого панкреатиту, що забезпечує прецизійний вплив на ключові ланки патогенезу. Огляд підтверджує високий потенціал нанопрепаратів у підвищенні ефективності та безпеки терапії ГП. Водночас подальші масштабні доклінічні дослідження і клінічні випробування необхідні для успішного впровадження цих нанотехнологічних підходів у клінічну практику.
Background. Acute pancreatitis (AP) is an acute inflammatory condition of the pancreas with no specific therapy. Current pharmacological interventions (anti-inflammatory, antioxidant) have limited efficacy due to low bioavailability, and short half-life. Innovative therapies are needed for targeted drug delivery to the pancreas with minimal side effects. Nanotechnology has emerged as a promising approach to this targeted therapy: nanomaterials can carry drugs to inflamed tissue and reduce off-target toxicity. Objective: analysis of current research on nanotechnology-based therapies for AP. Materials and methods. We systematically searched PubMed, Scopus, and Google Scholar (PRISMA 2020 guidelines). Out of 316 identified articles, 62 studies (mostly preclinical on animal models) met the inclusion criteria for nanotechnology-based therapeutic strategies in AP. Results. The included studies covered a broad range of nanotherapeutic approaches for AP, including polymeric, lipid-based, carbon, organometallic, and biomimetic nanocarriers. These nanoformulations enabled targeted drug delivery, controlled release, improved bioavailability, and reduced systemic toxicity. In experimental AP models, nanotherapies exhibited potent anti-inflammatory and antioxidant effects, including reduced inflammatory cytokines, scavenging of reactive oxygen species, stabilization of mitochondrial function and protection of acinar cells from injury. Overall, nanotherapeutic strategies improved outcomes in AP models and often outperformed standard treatment. Conclusions. Nanotherapy is a promising approach for precise targeting of key pathogenic pathways in AP. This review confirms the significant potential of nanotechnology to improve the efficacy and safety of AP treatment. However, further preclinical and clinical studies are needed to translate these nanotherapeutic approaches into clinical practice.
гострий панкреатит; нанотехнології; наночастинки; таргетна терапія; наномедицина
acute pancreatitis; nanotechnology; nanoparticles; targeted therapy; nanomedicine
Для ознакомления с полным содержанием статьи необходимо оформить подписку на журнал.
- Basile G, Vacante M, Corsaro A, Evola FR, Maugeri G, Barchitta M, et al. Treatment of acute pancreatitis. Minerva Surg. 2025;80(3):236-257. doi: 10.23736/S2724-5691.25.10773-9.
- Dupont B, Lozac’h J, Alves A. Etiological treatment of gallstone acute pancreatitis. World J Gastrointest Surg. 2025;17(5):105410. doi: 10.4240/wjgs.v17.i5.105410.
- Hamesch K, Hollenbach M, Guilabert L, Lahmer T, Koch A. Practical management of severe acute pancreatitis. Eur J Intern Med. 2025;133:1-13. doi: 10.1016/j.ejim.2024.10.030.
- Luo M, Jin T, Fang Y, Chen F, Zhu L, et al. Signaling Pathways Involved in Acute Pancreatitis. J Inflamm Res. 2025;18:2287-2303. doi: 10.2147/JIR.S485804.
- Zhang X, Xu C, Ji L, Zhang H. Endoplasmic reticulum stress in acute pancreatitis: Exploring the molecular mechanisms and therapeutic targets. Cell Stress Chaperones. 2025;30(3):119-129. doi: 10.1016/j.cstres.2025.03.001.
- Zhu L, Xu Y, Lei J. Molecular mechanism and potential role of mitophagy in acute pancreatitis. Mol Med. 2024;30(1):136. doi: 10.1186/s10020-024-00903-x.
- Chen X, Zhong R, Hu B. Mitochondrial dysfunction in the pathogenesis of acute pancreatitis. Hepatobiliary Pancreat Dis Int. 2025;24(1):76-83. doi: 10.1016/j.hbpd.2023.12.008.
- Sastre J, Pérez S, Sabater L, Rius-Pérez S. Redox signaling in the pancreas in health and disease. Physiol Rev. 2025;105(2):593-650. doi: 10.1152/physrev.00044.2023.
- Mihoc T, Latcu SC, Secasan CC, Dema V, Cumpanas AA, Selaru M, et al. Pancreatic Morphology, Immunology, and the Pathogenesis of Acute Pancreatitis. Biomedicines. 2024;12(11):2627. doi: 10.3390/biomedicines12112627.
- Beij A, Verdonk RC, van Santvoort HC, de-Madaria E, Voermans RP. Acute Pancreatitis: An Update of Evidence-Based Management and Recent Trends in Treatment Strategies. United European Gastroenterol J. 2025;13(1):97-106. doi: 10.1002/ueg2.12743.
- Guilabert L, Cárdenas-Jaén K, de-Madaria E. Initial Management of Acute Pancreatitis. Gastroenterol Clin North Am. 2025;54(1):21-36. doi: 10.1016/j.gtc.2024.07.001.
- Mittal N, Oza VM, Muniraj T, Kothari TH. Diagnosis and Management of Acute Pancreatitis. Diagnostics (Basel). 2025;15(3):258. doi: 10.3390/diagnostics15030258.
- Maatman TK, Zyromski NJ. Surgical Step-Up Approach in Management of Necrotizing Pancreatitis. Gastroenterol Clin North Am. 2025;54(1):53-74. doi: 10.1016/j.gtc.2024.10.001.
- Jiang X, Zheng YW, Bao S, Zhang H, Chen R, et al. Drug discovery and formulation development for acute pancreatitis. Drug Deliv. 2020;27(1):1562-1580. doi: 10.1080/10717544.2020.1840665.
- Ding L, Jian L, Xu J, He Q, Wang Y, Sun C, et al. Pharmacological Interventions for Acute Pancreatitis in Adults: An Overview of Systematic Reviews. J Evid Based Med. 2025;18(1):e70007. doi: 10.1111/jebm.70007.
- Jia W, Xu L, Xu W, Yang M, Zhang Y. Application of nanotechnology in the diagnosis and treatment of acute pancreatitis. Nanoscale Adv. 2022;4(8):1949-1961. doi: 10.1039/d2na00020b.
- Du W, Wang X, Zhou Y, Wu W, Huang H, Jin Z. From micro to macro, nanotechnology demystifies acute pancreatitis: a new generation of treatment options emerges. J Nanobiotechnology. 2025;23(1):57. doi: 10.1186/s12951-025-03106-6.
- Zheng X, Zhao J, Wang S, Hu L. Research Progress of Antioxidant Nanomaterials for Acute Pancreatitis. Molecules. 2022;27(21):7238. doi: 10.3390/molecules27217238.
- Zhang Q, Li S, Yu Y, Zhu Y, Tong R. A Mini-Review of Diagnostic and Therapeutic Nano-Tools for Pancreatitis. Int J Nanomedicine. 2022;17:4367-4381. doi: 10.2147/IJN.S385590.
- Liu L, Zhang Y, Li X, Deng J. Microenvironment of pancreatic inflammation: calling for nanotechnology for diagnosis and treatment. J Nanobiotechnology. 2023;21(1):443. doi: 10.1186/s12951-023-02200-x.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi: 10.1136/bmj.n71.
- Yang L, Liu X, Yang J, Wang K, Ai Z, et al. Biomimetic delivery of emodin via macrophage membrane-coated UiO-66-NH2 nanoparticles for acute pancreatitis treatment. Biochem Biophys Res Commun. 2024;702:149649. doi: 10.1016/j.bbrc.2024.149649.
- Abdel-Hakeem EA, Abdel-Hamid HA, Abdel Hafez SMN. The possible protective effect of Nano-Selenium on the endocrine and exocrine pancreatic functions in a rat model of acute pancreatitis. J Trace Elem Med Biol. 2020;60:126480. doi: 10.1016/j.jtemb.2020.126480.
- Hassanzadeh P, Arbabi E, Rostami F. Coating of ferulic acid-loaded silk fibroin nanoparticles with neutrophil membranes: A promising strategy against the acute pancreatitis. Life Sci. 2021;270:119128. doi: 10.1016/j.lfs.2021.119128.
- Zhou X, Cao X, Tu H, Zhang ZR, Deng L. Inflammation-Targeted Delivery of Celastrol via Neutrophil Membrane-Coated Nanoparticles in the Management of Acute Pancreatitis. Mol Pharm. 2019;16(3):1397-1405. doi: 10.1021/acs.molpharmaceut.8b01342.
- Yao Q, Jiang X, Zhai YY, Luo LZ, Xu HL, Xiao J, et al. Protective effects and mechanisms of bilirubin nanomedicine against acute pancreatitis. J Control Release. 2020;322:312-325. doi: 10.1016/j.jconrel.2020.03.034.
- Lin J, Wei Y, Gu X, Liu M, Wang M, Zhou R, et al. Nanotherapeutics-mediated restoration of pancreatic homeostasis and intestinal barrier for the treatment of severe acute pancreatitis. J Control Release. 2025;377:93-105. doi: 10.1016/j.jconrel.2024.11.022.
- Yan J, Liu H, Xu Y, Sun X, Meng X, Wei S, et al. Tailored “Three-stage booster” nano-extinguisher for synergistic treatment of severe acute pancreatitis by rectifying mitochondrial dysfunction and inhibiting pancreatic autodigestion. Acta Biomater. 2025;200:569-590. doi: 10.1016/j.actbio.2025.05.049.
- Abozaid OAR, Moawed FSM, Ahmed ESA, Ibrahim ZA. Cinnamic acid nanoparticles modulate redox signal and inflammatory response in gamma irradiated rats suffering from acute pancreatitis. Biochim Biophys Acta Mol Basis Dis. 2020;1866(11):165904. doi: 10.1016/j.bbadis.2020.165904.
- Wen E, Tian Y, Fang M, Zhang Y, Zhao H, Wang Z, et al. The P2X7-Mediated Mitochondrial ROS as an Emerging Core Target of Tuftsin Nanoparticles in Severe Acute Pancreatitis Therapy via Regulating Mitophagy. ACS Appl Mater Interfaces. 2025;17(5):7521-7538. doi: 10.1021/acsami.4c21010.
- Li J, Ye E, Huang J, Xu M, Zhang J, Zhang J, et al. Cysteine-modified PEGylated nanoparticles for targeted delivery of methylprednisolone to pancreatitis. Eur J Pharm Biopharm. 2024;195:114179. doi: 10.1016/j.ejpb.2024.114179.
- Song H, Zhang J, Lou N, Jiang X, Cui Y, Liu J, et al. Emodin nanocapsules inhibit acute pancreatitis by regulating lipid metabolic reprogramming in macrophage polarization. Phytomedicine. 2024;130:155763. doi: 10.1016/j.phymed.2024.155763.
- Khurana A, Anchi P, Allawadhi P, Kumar V, Sayed N, et al. Yttrium oxide nanoparticles reduce the severity of acute pancreatitis caused by cerulein hyperstimulation. Nanomedicine. 2019;18:54-65. doi: 10.1016/j.nano.2019.02.018.
- Wu J, Huang H, Xu W, Cui B, Sun P, Hao X, et al. Inflammation-driven biomimetic nano-polyphenol drug delivery system alleviates severe acute pancreatitis by inhibiting macrophage PANoptosis and pancreatic enzymes oversecretion. J Adv Res. 2026 Jan;79:783-789. doi: 10.1016/j.jare.2025.04.006. Online ahead of print.
- Shu J, Liao Y, Wang J, Zhang Y, Zhou W, Zhang H. Synthesis of Selenium Nanoparticles and Their Effect on Pancreatic Functions and Acute Pancreatitis in Rats. J Oleo Sci. 2024;73(3):351-358. doi: 10.5650/jos.ess23190.
- Fan JJ, Mei QX, Deng GY, Huang ZH, Fu Y, Hu JH, et al. Porous SiO2-coated ultrasmall selenium particles nanospheres attenuate cerulein-induce acute pancreatitis in mice by downregulating oxidative stress. J Dig Dis. 2021;22(6):363-372. doi: 10.1111/1751-2980.12989.
- Xie P, Zhang L, Shen H, Wu H, Zhao J, et al. Biodegra–dable MoSe2-polyvinylpyrrolidone nanoparticles with multi-enzyme activity for ameliorating acute pancreatitis. J Nanobiotechnology. 2022;20(1):113. doi: 10.1186/s12951-022-01288-x.
- Wen E, Cao Y, He S, Zhang Y, You L, Wang T, et al. The mitochondria-targeted Kaempferol nanoparticle ameliorates severe acute pancreatitis. J Nanobiotechnology. 2024;22(1):148. doi: 10.1186/s12951-024-02439-y.
- Zhang Q, Zhou J, Zhou J, Fang RH, Gao W, Zhang L. Lure-and-kill macrophage nanoparticles alleviate the severity of experimental acute pancreatitis. Nat Commun. 2021;12(1):4136. doi: 10.1038/s41467-021-24447-4.
- Awadeen RH, Boughdady MF, Zaghloul RA, Elsaed WM, Abu Hashim II, Meshali MM. Formulation of lipid polymer hybrid nanoparticles of the phytochemical Fisetin and its in vivo assessment against severe acute pancreatitis. Sci Rep. 2023;13(1):19110. doi: 10.1038/s41598-023-46215-8.
- Chen Y, Tao H, Chen R, Pan Y, Wang J, Gao R, et al. Biomimetic Nanoparticles Loaded with Ulinastatin for the Targeted Treatment of Acute Pancreatitis. Mol Pharm. 2023;20(8):4108-4119. doi: 10.1021/acs.molpharmaceut.3c00238.
- Chen G, Huang Y, Yu H, Wang J, Li H, Shen S, et al. Nanoparticles Fueled by Enzyme for the Treatment of Hyperlipidemic Acute Pancreatitis. ACS Biomater Sci Eng. 2024;10(11):7176-7190. doi: 10.1021/acsbiomaterials.4c00474.
- Fu Z, Wang D, Zheng C, Xie M, Chen Y, Zhou Y, et al. Elimination of intracellular Ca2+ overload by BAPTA-AM liposome nanoparticles: A promising treatment for acute pancreatitis. Int J Mol Med. 2024;53(4):34. doi: 10.3892/ijmm.2024.5358.
- Jin W, Xie X, Shen S, Zhou X, Wang S, et al. Ultrasmall polyvinylpyrrolidone-modified iridium nanoparticles with antioxidant and anti-inflammatory activity for acute pancreatitis alleviation. J Biomed Mater Res A. 2024;112(7):988-1003. doi: 10.1002/jbm.a.37679.
- Liu H, Liu S, Song X, Jiang A, Zou Y, Deng Y, et al. Nanoparticle encapsulated CQ/TAM combination harmonizes with MSCs in arresting progression of severity in AP mice through iNOS (IDO) signaling. Mater Today Bio. 2022;14:100226. doi: 10.1016/j.mtbio.2022.100226.
- Shi F, Ergashev A, Pan Z, Sun H, Kong L, Jin Y, et al. Macrophage-mimicking nanotherapy for attenuation of acute pancreatitis. Mater Today Bio. 2024;30:101406. doi: 10.1016/j.mtbio.2024.101406.
- Wang L, Gao Z, Tian M, Liu L, Xie J, Chen M, et al. A Nanosystem Alleviates Severe Acute Pancreatitis via Reactive Oxygen Species Scavenging and Enhancing Mitochondrial Autophagy. Nano Lett. 2025;25(21):8644-8654. doi: 10.1021/acs.nanolett.5c01495.
- Khurana A, Anchi P, Allawadhi P, Kumar V, Sayed N, et al. Superoxide dismutase mimetic nanoceria restrains cerulein induced acute pancreatitis. Nanomedicine (Lond). 2019;14(14):1805-1825. doi: 10.2217/nnm-2018-0318.
- Zhang Q, Shen Y, Zhang C, Zhang H, Li X, Yang S, et al. Immunoengineered mitochondria for efficient therapy of acute organ injuries via modulation of inflammation and cell repair. Sci Adv. 2025;11(12):eadj1896. doi: 10.1126/sciadv.adj1896.
- Wang Y, Qian D, Wang X, Zhang X, Li Z, Meng X, et al. Biomimetic Trypsin-Responsive Structure-Bridged Mesoporous Organosilica Nanomedicine for Precise Treatment of Acute Pancreatitis. ACS Nano. 2024;18(29):19283-19302. doi: 10.1021/acsnano.4c05369.
- Wang Y, Wang X, Zhang X, Zhang B, Meng X, Qian D, et al. Inflammation and Acinar Cell Dual-Targeting Nanomedicines for Synergistic Treatment of Acute Pancreatitis via Ca2+ Homeostasis Regulation and Pancreas Autodigestion Inhibition. ACS Nano. 2024;18(18):11778-11803. doi: 10.1021/acsnano.4c00218.
- Wang D, Wang S, Liu J, Shi X, Xiong T, Li R, et al. Nanomedicine Penetrating Blood-Pancreas Barrier for Effective Treatment of Acute Pancreatitis. Adv Sci (Weinh). 2025;12(13):e2413925. doi: 10.1002/advs.202413925.
- Nagao S, Taguchi K, Sakai H, Yamasaki K, Watanabe H, et al. Carbon monoxide-bound hemoglobin vesicles ameliorate multiorgan injuries induced by severe acute pancreatitis in mice by their anti-inflammatory and antioxidant properties. Int J Nanomedicine. 2016;11:5611-5620. doi: 10.2147/IJN.S118185.
- Chen K, Zhang Z, Fang Z, Zhang J, Liu Q, Dong W, et al. Aged-Signal-Eliciting Nanoparticles Stimulated Macrophage-Mediated Programmed Removal of Inflammatory Neutrophils. ACS Nano. 2023;17(14):13903-13916. doi: 10.1021/acsnano.3c03815.
- Mei Q, Deng G, Huang Z, Yin Y, Li C, Hu J, et al. Porous COS@SiO2 Nanocomposites Ameliorate Severe Acute Pancreatitis and Associated Lung Injury by Regulating the Nrf2 Signaling Pathway in Mice. Front Chem. 2020;8:720. doi: 10.3389/fchem.2020.00720.
- Li Q, Cao Q, Yuan Z, Wang M, Chen P, Wu X. A novel self-nanomicellizing system of empagliflozin for oral treatment of acute pancreatitis: An experimental study. Nanomedicine. 2022;42:102534. doi: 10.1016/j.nano.2022.102534.
- Yang Q, Luo Y, Ge P, Lan B, Liu J, Wen H, et al. Emodin Ameliorates Severe Acute Pancreatitis-Associated Acute Lung Injury in Rats by Modulating Exosome-Specific miRNA Expression Profiles. Int J Nanomedicine. 2023;18:6743-6761. doi: 10.2147/IJN.S428924.
- Lopez-Pascual A, Santamaria E, Ardaiz N, Uriarte I, Pal–mer T, Graham AR, et al. FGF21 and APOA1 mRNA-based therapies for the treatment of experimental acute pancreatitis. J Transl Med. 2025;23(1):122. doi: 10.1186/s12967-025-06129-7.
- Lu X, Gao Z, Yu Y, Zhang L, Huang J, Zhang X, et al. Natural product-based nano-antioxidant for the treatment of acute pancreatitis. Regen Biomater. 2025;12:rbaf012. doi: 10.1093/rb/rbaf012.
- Zou Y, Qin Q, Song X, Deng Y, Liu S, Liu H, et al. The potency of aloe emodin-loaded nanoparticles in conjunction with IFN-γ for the pretreatment of mesenchymal stem cells with class II transactivator silence to alleviate severe acute pancreatitis. MedComm — Biomater Appl. 2025;4:e70001. doi: 10.1002/mba2.70001.
- Xie X, Zhao J, Gao W, Chen J, Hu B, et al. Prussian blue nanozyme-mediated nanoscavenger ameliorates acute pancreatitis via inhibiting TLRs/NF-κB signaling pathway. Theranostics. 2021;11(7):3213-3228. doi: 10.7150/thno.52010.
- Taguchi K, Nagao S, Maeda H, Yanagisawa H, Sakai H, Yamasaki K, et al. Biomimetic carbon monoxide delivery based on hemoglobin vesicles ameliorates acute pancreatitis in mice via the regulation of macrophage and neutrophil activity. Drug Deliv. 2018;25(1):1266-1274. doi: 10.1080/10717544.2018.1477860.
- Dang SC, Zeng YH, Wang PJ, Chen BD, Chen RF, Kumar Singh A, et al. Clodronate-superparamagnetic iron oxide-containing liposomes attenuate renal injury in rats with severe acute pancreatitis. J Zhejiang Univ Sci B. 2014;15(6):556-565. doi: 10.1631/jzus.B1300244.
- Khaksar MR, Rahimifard M, Baeeri M, Maqbool F, Navaei-Nigjeh M, Hassani S, et al. Protective effects of cerium oxide and yttrium oxide nanoparticles on reduction of oxidative stress induced by sub-acute exposure to diazinon in the rat pancreas. J Trace Elem Med Biol. 2017;41:79-90. doi: 10.1016/j.jtemb.2017.02.013.
- Rashidian A, Ghafari H, Chamanara M, Dehpour AR, Muhammadnejad A, Akbarian R, et al. The protective effect of nano-Curcumin in experimental model of acute pancreatitis: The involvement of TLR4/NF-kB pathway. Nanomed J. 2018;5(3):138-143. doi: 10.22038/nmj.2018.005.0003.
- Shahin NN, Shamma RN, Ahmed IS. A Nano-Liposomal Formulation of Caffeic Acid Phenethyl Ester Modulates Nrf2 and –NF-κβ Signaling and Alleviates Experimentally Induced Acute Pancreatitis in a Rat Model. Antioxidants (Basel). 2022;11(8):1536. doi: 10.3390/antiox11081536.
- Karole A, Dinakar YH, Sagar P, Parvez S, Kumar R, Mudavath SL. Targeting inflammatory macrophages with tailored bioactive β-glucan conjugated polymeric nanoparticles for the treatment of pancreatitis. Chemical Engineering Journal. 2025;516:163838. doi: 10.1016/j.cej.2025.163838.
- Zhang L, Xie P, Wu H, Zhao J, Wang S. 2D MoSe2@PVP nanosheets with multi-enzyme activity alleviate the acute pancreatitis via scavenging the reactive oxygen and nitrogen species. Chemical Engineering Journal. 2022;446:136792. doi: 10.1016/j.cej.2022.1367.
- Tang Y, Han Y, Liu L, Shen W, Zhang H, Wang Y, et al. Protective effects and mechanisms of G5 PAMAM dendrimers against acute pancreatitis induced by caerulein in mice. Biomacromolecules. 2015;16(1):174-182. doi: 10.1021/bm501390d.
- Wang Y, Li Y, Gao S, Yu X, Chen Y, Lin Y. Tetrahedral Framework Nucleic Acids Can Alleviate Taurocholate-Induced Severe Acute Pancreatitis and Its Subsequent Multiorgan Injury in Mice. Nano Lett. 2022;22(4):1759-1768. doi: 10.1021/acs.nanolett.1c05003.
- Chuang EY, Lin KJ, Huang TY, Chen HL, Miao YB, Lin PY, et al. An Intestinal “Transformers”-like Nanocarrier System for Enhancing the Oral Bioavailability of Poorly Water-Soluble Drugs. ACS Nano. 2018;12(7):6389-6397. doi: 10.1021/acsnano.8b00470.
- Zhang J, Guo M, He Q, Zhang Z, Wu B, Wu H, et al. Precise Control of Metal Active Sites of Metal-Organic Framework Nanozymes for Achieving Excellent Enzyme-Like Activity and Efficient Pancreatitis Therapy. Small. 2024;20(32):e2310675. doi: 10.1002/smll.202310675.
- Jing F, Zhu Y, Li F, Wang Y, Yu X, Zhang K, et al. Bimetallic ions-doped carbon dots nanotheranostics for imaging-guided macrophage polarization/ROS scavenging in acute pancreatitis. Chemical Engineering Journal. 2023;465:142675. doi: 10.1016/j.cej.2023.142675.
- Wang L, Tian M, Dong B, Li W, Shi L, Tong Y, et al. Macrophage-targeted emodin nanomaterials for effective acute pancreatitis treatment via modulation of the JNK pathway. Biomater Sci. 2025;13(16):4461-4481. doi: 10.1039/d5bm00018a.
- Chegini M, Sadeghi A, Zaeri F, Zamani M, Hekmatdoost A. Nano-curcumin supplementation in patients with mild and moderate acute pancreatitis: A randomized, placebo-controlled trial. Phytother Res. 2023;37(11):5279-5288. doi: 10.1002/ptr.7958.
- Farghly G, Hanafy N, El-Tonsy M, Farid N. Protective Effect of Nano-Moringa oleifera leaves Extract and/or Low Doses of γ-Irradiation on Acute Pancreatitis Induced in Rats Model. International Journal of Theoretical and Applied Research. 2023;2(2):215-224. doi: 10.21608/IJTAR.2023.205193.1052.
- Li Y, Cai R, Chen K, Zhang Y, Chen X, Sun B, et al. Ca/Fe-Based Nanozymes Relieve Severe Acute Pancreatitis by Ferroptosis Regulation and Reactive Oxygen Species Scavenging. ACS Appl Nano Mater. 2023;6(14):12968-12979. doi: 10.1021/acsanm.3c01697.
- Bao X, Xin G, Zhou Q, Wang T, Xu X, Feng L, et al. Dual-scavenging Mn3O4-PEI nanoparticles targeting ROS and cfDNA for acute pancreatitis therapy. Nanomedicine. 2025;68:102848. doi: 10.1016/j.nano.2025.102848.
- Wu L, Cai R, Li Y, Liao S, Song Y, Li Y, et al. Modula–ting Macrophage Polarization for Severe Acute Pancreatitis The–rapy via Cisplatin-like Prussian Blue Nanozymes. Theranostics. 2025;15(17):8916-8934. doi: 10.7150/thno.113523.
- Ahmed NH, Mosleh M, Zahran F. Relieve acute pancreatitis in Wistar rats subjected to nano-formulated metformin drug with Chitosan and/or low doses γ-Irradiation. Discover Applied Sciences. 2025;7:1037. doi: 10.1007/s42452-025-07353-y.
- Abd El-hameed SM, El-Tonsy MM, Ahmed NH, Abdelhafez HM. The Possible Therapeutic Effect of Nanoparticles of Moringa oleifera Leaves Extract and\or Low Doses of Gamma Radiation on Liver Injury Induced in Rats by Acute Pancreatitis. Egypt Acad J Biolog Sci. 2025;17(2):25-53. doi: 10.21608/EAJBSZ.2025.449823.
- Jiang H, Xu Z, Song Q, Tao J, Liu J, Wang Q, et al. Targeting PFKFB3 to restore glucose metabolism in acute pancreatitis via nanovesicle delivery. Mol Med. 2025;31(1):253. doi: 10.1186/s10020-025-01261-y.
- Liu J, Zeng L, Shen Q, Yu T, Ren Y, Cao X, et al. Endocrine-driven FGF21 mRNA therapy via route-adaptable lipopolymer nanoparticles for the treatment of pancreatitis. Chemical Engineering Journal. 2025;520:166411. doi: 10.1016/j.cej.2025.166411.