Журнал «Медицина неотложных состояний» Том 22, №3, 2026
Вернуться к номеру
Кишковий бар’єр як «файрвол» при гострому панкреатиті: дисбіоз і каскад ускладнень
Авторы: Чуклін С.М., Чуклін С.С.
Медичний центр Святої Параскеви, м. Львів, Україна
Рубрики: Медицина неотложных состояний
Разделы: Справочник специалиста
Версия для печати
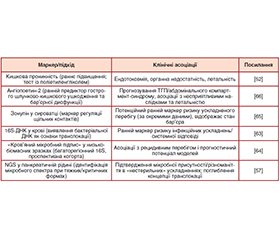
Актуальність. Гострий панкреатит (ГП) є гетерогенним системним запальним синдромом, у якому рання недостатність кишкового бар’єра та дисбіоз сприяють ендотоксемії, транслокації мікробних продуктів і ескалації системних ускладнень. Мета: узагальнити сучасні докази ролі кишкового бар’єра та мікробіоти у формуванні ускладнень ГП, окреслити механістичні ланки каскаду та потенціал багатоомних біомаркерів для ранньої стратифікації ризику. Матеріали та методи. Наративний синтез із елементами систематизованого пошуку. Пошук у PubMed/Medline, Scopus, Google Scholar; включали клінічні та експериментальні роботи з оцінкою мікробіоти/метаболітів і кінцевих точок (органна недостатність, інфікований панкреонекроз, сепсис, ГРДС, летальність). Результати. Дані свідчать про раннє підвищення кишкової проникності при ГП, що корелює з тяжкістю перебігу та створює «вікно» для дисбіозу і транслокації, особливо при некротизуючому фенотипі. Найбільш відтворювані таксономічні зсуви включають зростання Enterococcus/Enterobacteriaceae/Escherichia — Shigella та зниження Bifidobacterium/Blautia, асоційовані з інтенсивною терапією, інфекційними ускладненнями й легеневими проявами. Інтегративні підходи (метагеноміка + метаболоміка) виявляють сигнатури, пов’язані з ризиком органної недостатності/ГРДС, а функціональний внесок мікробіоти опосередковується метаболітами (SCFA, жовчні кислоти, ароматичні токсини), що підвищують інформативність прогностичних моделей за інтеграції з клінічними параметрами. Висновки. Кишковий бар’єр і мікробіота є ключовими детермінантами каскаду ускладнень ГП; для впровадження мікробіомних біомаркерів потрібні стандартизація преаналітики/аналітики, максимально ранній забір зразків, контроль конфаундерів і зовнішня валідація моделей.
Background. Acute pancreatitis (AP) is a heterogeneous systemic inflammatory syndrome in which early failure of the intestinal barrier and gut dysbiosis promote endotoxemia, translocation of microbial products, and increased systemic complications. Objective: to synthesize current evidence on the role of the gut barrier and microbiota in AP complications, outline key mechanistic links, and highlight how multi-omics biomarkers may support early risk stratification. Materials and methods. Narrative synthesis with a structured search. PubMed/Medline, Scopus, and Google Scholar were searched. We included clinical and experimental studies assessing microbiota/metabolites and relevant outcomes (organ failure, infected pancreatic necrosis, sepsis, acute respiratory distress syndrome, mortality). Results. Evidence indicates an early rise in intestinal permeability in AP, associated with disease severity and enabling dysbiosis and microbial translocation, particularly in necrotizing phenotypes. The most reproducible taxonomic shifts include increases in Enterococcus/Enterobacteriaceae/Escherichia-Shigella and decreases in Bifidobacterium/Blautia, which in several cohorts correlate with intensive care need, infectious complications, and pulmonary involvement. Integrative approaches (metagenomics + metabolomics) reveal signatures linked to the risk of organ failure/acute respiratory distress syndrome, while microbiota-derived metabolites (short-chain fatty acids, bile acids, aromatic toxins) provide a functional layer that can improve prognostic models when combined with clinical variables. Conclusions. The gut barrier and microbiota are pivotal drivers of the AP complication cascade. Clinical translation of microbiome-based biomarkers requires standardized preanalytics/analytics, very early sampling, rigorous confounder control, and external validation on independent cohorts.
гострий панкреатит; кишковий бар’єр; мікробіота; дисбіоз; транслокація; ендотоксемія; багатоомні біомаркери
acute pancreatitis; intestinal barrier; microbiota; dysbiosis; translocation; endotoxemia; multi-omics biomarkers
Для ознакомления с полным содержанием статьи необходимо оформить подписку на журнал.
- Huang Y, Badurdeen DS. Acute Pancreatitis Review. Turk J Gastroenterol. 2023 Aug;34(8):795-801. doi: 10.5152/tjg.2023.23175.
- Hamesch K, Hollenbach M, Guilabert L, Lahmer T, Koch A. Practical management of severe acute pancreatitis. Eur J Intern Med. 2025 Mar;133:1-13. doi: 10.1016/j.ejim.2024.10.030.
- Banks PA, Bollen TL, Dervenis C, et al. Classification of acute pancreatitis — 2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013 Jan;62(1):102-111. doi: 10.1136/gutjnl-2012-302779.
- IAP/APA/EPC/IPC/JPS Working Group. International Association of Pancreatology Revised Guidelines on Acute Pancreatitis 2025: Supported and Endorsed by the American Pancreatic Association, European Pancreatic Club, Indian Pancreas Club, and Japan Pancreas Society. Pancreatology. 2025 Sep;25(6):770-814. doi: 10.1016/j.pan.2025.04.020.
- Li Y, Li J, Li S, et al. Exploring the gut microbiota's crucial role in acute pancreatitis and the novel therapeutic potential of derived extracellular vesicles. Front Pharmacol. 2024 Jul 26;15:1437894. doi: 10.3389/fphar.2024.1437894.
- Wu J-L, Luo J-Y, Deng X-Y, Lau TY, Jiang Z-B. The role of composition of gut microbiota in reflecting the severity of acute pancreatitis is probably overstated. Microbiol Spectr. 2024 May 2;12(5):e0018424. doi: 10.1128/spectrum.00184-24.
- Agarwal S, Goswami P, Poudel S, et al. Acute pancreatitis is characterized by generalized intestinal barrier dysfunction in early stage. Pancreatology. 2023 Jan;23(1):9-17. doi: 10.1016/j.pan.2022.11.011.
- Pan L, Yin N, Duan M, Mei Q, Zeng Y. The role of gut microbiome and its metabolites in pancreatitis. mSystems. 2024 Oct 22;9(10):e0066524. doi: 10.1128/msystems.00665-24.
- Lei Y, Tang L, Liu S, et al. Parabacteroides produces acetate to alleviate heparanase-exacerbated acute pancreatitis through redu–cing neutrophil infiltration. Microbiome. 2021 May 20;9(1):115. doi: 10.1186/s40168-021-01065-2.
- Lange R, Glaubitz J, Frost F, et al. Examination of duodenal and colonic microbiome changes in mouse models of acute and chronic pancreatitis. Sci Rep. 2024 Oct 21;14(1):24754. doi: 10.1038/s41598-024-75564-1.
- Jing H, Chang Q, Xu Y, et al. Effect of aging on acute pancreatitis through gut microbiota. Front Microbiol. 2022 Jul 28;13:897992. doi: 10.3389/fmicb.2022.897992.
- Ammer-Herrmenau C, Antweiler KL, Asendorf T, et al. Gut microbiota predicts severity and reveals novel metabolic signatures in acute pancreatitis. Gut. 2024 Feb 23;73(3):485-495. doi: 10.1136/gutjnl-2023-330987.
- Nie HY, Ge J, Huang GX, et al. New insights into the intestinal barrier through “gut-organ” axes and a glimpse of the microgravityʼs effects on intestinal barrier. Front Physiol. 2024 Oct 10;15:1465649. doi: 10.3389/fphys.2024.1465649.
- Chen H, Wang Y, Zippi M, Fiorino S, Hong W. Oxidative stress, DAMPs, and immune cells in acute pancreatitis: molecular mechanisms and therapeutic prospects. Front Immunol. 2025 Aug 20;16:1608618. doi: 10.3389/fimmu.2025.1608618.
- Mattke J, Darden CM, Lawrence MC, et al. Toll-like receptor 4 in pancreatic damage and immune infiltration in acute pancreatitis. Front Immunol. 2024 Mar 22;15:1362727. doi: 10.3389/fimmu.2024.1362727.
- Zhang Z, Tanaka I, Pan Z, Ernst PB, Kiyono H, Kurashima Y. Intestinal homeostasis and inflammation: Gut microbiota at the crossroads of pancreas-intestinal barrier axis. Eur J Immunol. 2022 Jul;52(7):1035-1046. doi: 10.1002/eji.202149532.
- Wang Z, Liu J, Li F, et al. The gut-lung axis in severe acute pancreatitis-associated lung injury: The protection by the gut microbiota through short-chain fatty acids. Pharmacol Res. 2022 Aug;182:106321. doi: 10.1016/j.phrs.2022.106321.
- Liu Q, Zhu X, Guo S. From pancreas to lungs: The role of immune cells in severe acute pancreatitis and acute lung injury. Immun Inflamm Dis. 2024 Jul;12(7):e1351. doi: 10.1002/iid3.1351.
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021 Mar 29;372:n71. doi: 10.1136/bmj.n71.
- Liu Q, Ruan K, An Z, et al. Updated review of research on the role of the gut microbiota and microbiota-derived metabolites in acute pancreatitis progression and inflammation-targeted therapy. Int J Biol Sci. 2025 Jan 20;21(3):1242-1258. doi: 10.7150/ijbs.108858.
- Du Z-C, Li G-Q, Luo Y, Bai X-W, Sun B. The role of gut microbiota in acute pancreatitis: new perspectives in pathogenesis and therapeutic approaches. J Pancreatol. 2024;7:61-71. doi: 10.1097/JP9.0000000000000131.
- Liu J, Yan Q, Li S, et al. Integrative metagenomic and metabolomic analyses reveal the potential of gut microbiota to exacerbate acute pancreatitis. NPJ Biofilms Microbiomes. 2024 Mar 21;10(1):29. doi: 10.1038/s41522-024-00499-4.
- Wang Z, Guo M, Li J, et al. Composition and functional profiles of gut microbiota reflect the treatment stage, severity, and etiology of acute pancreatitis. Microbiol Spectr. 2023 Sep 12;11(5):e0082923. doi: 10.1128/spectrum.00829-23.
- Hu X, Han Z, Zhou R, et al. Altered gut microbiota in the early stage of acute pancreatitis were related to the occurrence of acute respiratory distress syndrome. Front Cell Infect Microbiol. 2023 Mar 6;13:1127369. doi: 10.3389/fcimb.2023.1127369.
- Li F, Wang Z, Cao Y, et al. Intestinal Mucosal Immune Barrier: A Powerful Firewall Against Severe Acute Pancreatitis-Associated Acute Lung Injury via the Gut-Lung Axis. J Inflamm Res. 2024 Apr 10;17:2173-2193. doi: 10.2147/JIR.S448819.
- Zhou F, Liu Y, Shi Y, Wu N, Xie Y, Zhou X. Association between gut microbiota and acute pancreatitis: a bidirectional Mendelian randomization study. J Gastroenterol Hepatol. 2024 Sep;39(9):1895-1902. doi: 10.1111/jgh.16658.
- Nan B, Jin L, Wang T, et al. Correlation between gut microbiota and pancreatitis: a bidirectional Mendelian randomization. Eur J Gastroenterol Hepatol. 2025 Jan 1;37(1):62-69. doi: 10.1097/MEG.0000000000002861.
- Deng YY, Shamoon M, He Y, Bhatia M, Sun J. Cathelicidin-related antimicrobial peptide modulates the severity of acute pancreatitis in mice. Mol Med Rep. 2016 May;13(5):3881-3885. doi: 10.3892/mmr.2016.5008.
- Fu Y, Mei Q, Yin N, et al. Paneth Cells Protect against Acute Pancreatitis via Modulating Gut Microbiota Dysbiosis. mSystems. 2022 Jun 28;7(3):e0150721. doi: 10.1128/msystems.01507-21.
- Li XY, He C, Zhu Y, Lu NH. Role of gut microbiota on intestinal barrier function in acute pancreatitis. World J Gastroenterol. 2020 May 14;26(18):2187-2193. doi: 10.3748/wjg.v26.i18.2187.
- Wang Z, Liu J, Wang Y, et al. Identification of Key Biomar–kers Associated with Immunogenic Cell Death and Their Regulatory Mechanisms in Severe Acute Pancreatitis Based on WGCNA and Machine Learning. Int J Mol Sci. 2023 Feb 3;24(3):3033. doi: 10.3390/ijms24033033.
- Wu L, Hu J, Yi X, et al. Gut microbiota interacts with inflammatory responses in acute pancreatitis. Therap Adv Gastroenterol. 2023 Oct 10;16:17562848231202133. doi: 10.1177/17562848231202133.
- Xuan J-L, Zhu Y-W, Xu W-H, et al. Integrative effects of transcutaneous electrical acustimulation on abdominal pain, gastrointestinal motility, and inflammation in patients with early-stage acute pancreatitis. Neurogastroenterol Motil. 2022 Apr;34(4):e14249. doi: 10.1111/nmo.14249.
- Cui X, Guo H, Liu Z, et al. The intricate interplay between acute pancreatitis and small intestinal bacterial overgrowth: Unrave–ling the unknown. Clin Nutr. 2025 Aug;51:362-372. doi: 10.1016/j.clnu.2025.06.008.
- Kurashima Y, Kigoshi T, Murasaki S, et al. Pancreatic glycoprotein 2 is a first line of defense for mucosal protection in intestinal inflammation. Nat Commun. 2021 Feb 16;12(1):1067. doi: 10.1038/s41467-021-21277-2.
- Murakami M, Yamamoto K, Miki Y, Murase R, Sato H, Taketomi Y. The Roles of the Secreted Phospholipase A2 Gene Fa–mily in Immunology. Adv Immunol. 2016;132:91-134. doi: 10.1016/bs.ai.2016.05.001.
- Nishiyama H, Nagai T, Kudo M, et al. Supplementation of pancreatic digestive enzymes alters the composition of intestinal microbiota in mice. Biochem Biophys Res Commun. 2018 Jan 1;495(1):273-279. doi: 10.1016/j.bbrc.2017.10.130.
- Edogawa S, Edwinson AL, Peters SA, et al. Serine proteases as luminal mediators of intestinal barrier dysfunction and symptom severity in IBS. Gut. 2020 Jan;69(1):62-73. doi: 10.1136/gutjnl-2018-317416.
- Koduri RS, Grönroos JO, Laine VJ, et al. Bactericidal properties of human and murine groups I, II, V, X, and XII secreted phospholipases A(2). J Biol Chem. 2002 Feb 22;277(8):5849-5857. doi: 10.1074/jbc.M109699200.
- Chairatana P, Chu H, Castillo PA, Shen B, Bevins CL, Nolan EM. Proteolysis Triggers Self-Assembly and Unmasks Innate Immune Function of a Human α-Defensin Peptide. Chem Sci. 2016 Mar 1;7(3):1738-1752. doi: 10.1039/C5SC04194E.
- Li X, He C, Li N, et al. The interplay between the gut microbiota and NLRP3 activation affects the severity of acute pancreatitis in mice. Gut Microbes. 2020 Nov 1;11(6):1774-1789. doi: 10.1080/19490976.2020.1770042.
- Zheng J, Lou L, Fan J, et al. Commensal Escherichia coli Aggravates Acute Necrotizing Pancreatitis through Targeting of Intestinal Epithelial Cells. Appl Environ Microbiol. 2019 May 30;85(12):e00059-19. doi: 10.1128/AEM.00059-19.
- Jin M, Zhang H, Wu M, et al. Colonic interleukin-22 protects intestinal mucosal barrier and microbiota abundance in severe acute pancreatitis. FASEB J. 2022 Mar;36(3):e22174. doi: 10.1096/fj.202101371R.
- Xiong Y, Ji L, Zhao Y, Liu A, Wu D, Qian J. Sodium Bu–tyrate Attenuates Taurocholate-Induced Acute Pancreatitis by Maintaining Colonic Barrier and Regulating Gut Microorganisms in Mice. Front Physiol. 2022 Mar 17;13:813735. doi: 10.3389/fphys.2022.813735.
- Chen J, Wang Y, Shi Y, Liu Y, Wu C, Luo Y. Association of Gut Microbiota With Intestinal Ischemia/Reperfusion Injury. Front Cell Infect Microbiol. 2022 Jul 12;12:962782. doi: 10.3389/fcimb.2022.962782.
- Gao Y, Davis B, Zhu W, Zheng N, Meng D, Walker WA. Short-chain fatty acid butyrate, a breast milk metabolite, enhances immature intestinal barrier function genes in response to inflammation in vitro and in vivo. Am J Physiol Gastrointest Liver Physiol. 2021 Apr 1;320(4):G521-G530. doi: 10.1152/ajpgi.00279.2020.
- Wei YH, Ma X, Zhao JC, Wang XQ, Gao CQ. Succinate metabolism and its regulation of host-microbe interactions. Gut Microbes. 2023 Jan-Dec;15(1):2190300. doi: 10.1080/19490976.2023.2190300.
- Glaubitz J, Wilden A, Frost F, et al. Activated regulatory T-cells promote duodenal bacterial translocation into necrotic areas in severe acute pancreatitis. Gut. 2023 Jul;72(7):1355-1369. doi: 10.1136/gutjnl-2022-327448.
- Yu S, Xiong Y, Xu J, et al. Identification of Dysfunctional Gut Microbiota Through Rectal Swab in Patients with Different Severity of Acute Pancreatitis. Dig Dis Sci. 2020 Nov;65(11):3223-3237. doi: 10.1007/s10620-020-06061-4.
- Zou M, Yang Z, Fan Y, et al. Gut microbiota on admission as predictive biomarker for acute necrotizing pancreatitis. Front Immunol. 2022 Aug 29;13:988326. doi: 10.3389/fimmu.2022.988326.
- Liu J, Luo M, Qin S, Li B, Huang L, Xia X. Significant Succession of Intestinal Bacterial Community and Function During the Initial 72 Hours of Acute Pancreatitis in Rats. Front Cell Infect Microbiol. 2022 Apr 29;12:808991. doi: 10.3389/fcimb.2022.808991.
- Ammori BJ, Leeder PC, King RF, et al. Early increase in intestinal permeability in patients with severe acute pancreatitis: correlation with endotoxemia, organ failure, and mortality. J Gastrointest Surg. 1999 May-Jun;3(3):252-262. doi: 10.1016/s1091-255x(99)80067-5.
- Ahuja M, Schwartz DM, Tandon M, et al. Orai1-Mediated Antimicrobial Secretion from Pancreatic Acini Shapes the Gut Microbiome and Regulates Gut Innate Immunity. Cell Metab. 2017 Mar 7;25(3):635-646. doi: 10.1016/j.cmet.2017.02.007.
- Zhu Y, He C, Li X, et al. Gut microbiota dysbiosis worsens the severity of acute pancreatitis in patients and mice. J Gastroenterol. 2019 Apr;54(4):347-358. doi: 10.1007/s00535-018-1529-0.
- Gong L, Li X, Ji L, et al. Characterization and comparison of gut microbiota in patients with acute pancreatitis by metagenomics and culturomics. Heliyon. 2025 Jan 25;11(3):e42243. doi: 10.1016/j.heliyon.2025.e42243.
- Chmielarczyk A, Golińska E, Tomusiak-Plebanek A, et al. Microbial dynamics of acute pancreatitis: integrating culture, sequencing, and bile impact on bacterial populations and gaseous metabolites. Front Microbiol. 2025 Feb 12;16:1544124. doi: 10.3389/fmicb.2025.1544124.
- Sun N, Chen Y, Zhang J, et al. Identification and characte–rization of pancreatic infections in severe and critical acute pancreatitis patients using 16S rRNA gene next generation sequencing. Front Microbiol. 2023 Jun 14;14:1185216. doi: 10.3389/fmicb.2023.1185216.
- Li G, Liu L, Lu T, et al. Gut microbiota aggravates neutrophil extracellular traps-induced pancreatic injury in hypertriglyceridemic pancreatitis. Nat Commun. 2023 Oct 4;14(1):6179. doi: 10.1038/s41467-023-41950-y.
- Liu Y, Liu H, Rong Y, et al. Alterations of oral microbiota are associated with the development and severity of acute pancreatitis. J Oral Microbiol. 2023 Oct 5;15(1):2264619. doi: 10.1080/20002297.2023.2264619.
- Zhao Z, Han L, Tuerxunbieke B, et al. Effects of gut microbiota and metabolites on pancreatitis: a 2-sample Mendelian randomization study. J Gastrointest Surg. 2025 Feb;29(2):101885. doi: 10.1016/j.gassur.2024.101885.
- Wang H, Chen Y, Han Y, et al. Comparative study of gut microbiota and metabolite variations between severe and mild acute pancreatitis patients at different stages. Microb Pathog. 2025 Jan;198:107030. doi: 10.1016/j.micpath.2024.107030.
- Zhao MQ, Fan MY, Cui MY, et al. Profile of intestinal fungal microbiota in acute pancreatitis patients and healthy individuals. Gut Pathog. 2025 Jan 8;17(1):1. doi: 10.1186/s13099-024-00675-z.
- Li Q, Wang C, Tang C, He Q, Li N, Li J. Bacteremia in patients with acute pancreatitis as revealed by 16S ribosomal RNA gene-based techniques. Crit Care Med. 2013 Aug;41(8):1938-50. doi: 10.1097/CCM.0b013e31828a3dba.
- Wang QW, Zheng H, Yang Y, et al. Distinct microbial signatures and their predictive value in recurrent acute pancreatitis: insights from 5-region 16S rRNA gene sequencing. Front Immunol. 2025 Feb 28;16:1558983. doi: 10.3389/fimmu.2025.1558983.
- Avcıoğlu U, Eruzun H. Serum Zonulin Levels as an Early Biomarker in Predicting the Severity and Complications of Acute Pancreatitis. Eurasian J Med. 2023 Feb;55(1):78-82. doi: 10.5152/eurasianjmed.2022.0272.
- Huang Q, Wu Z, Chi C, et al. Angiopoietin-2 Is an Early Predictor for Acute Gastrointestinal Injury and Intestinal Barrier Dysfunction in Patients with Acute Pancreatitis. Dig Dis Sci. 2021 Jan;66(1):114-120. doi: 10.1007/s10620-020-06138-0.
- Heckler M, Hackert T, Hu K, Halloran CM, Büchler MW, Neoptolemos JP. Severe acute pancreatitis: surgical indications and treatment. Langenbecks Arch Surg. 2021 May;406(3):521-535. doi: 10.1007/s00423-020-01944-6.
- Podda M, Pellino G, Di Saverio S, et al. Infected pancreatic necrosis: outcomes and clinical predictors of mortality. A post hoc analysis of the MANCTRA-1 international study. Updates Surg. 2023 Apr;75(3):493-522. doi: 10.1007/s13304-023-01488-6.
- Ning C, Ouyang H, Shen D, et al. Prediction of survival in patients with infected pancreatic necrosis: a prospective cohort study. Int J Surg. 2024 Feb 1;110(2):777-787. doi: 10.1097/JS9.0000000000000844.
- Hu X, Gong L, Zhou R, et al. Variations in Gut Microbiome are Associated with Prognosis of Hypertriglyceridemia-Associated Acute Pancreatitis. Biomolecules. 2021 May 6;11(5):695. doi: 10.3390/biom11050695.
- Liang XY, Jia TX, Zhang M. Intestinal bacterial overgrowth in the early stage of severe acute pancreatitis is associated with acute respiratory distress syndrome. World J Gastroenterol. 2021 Apr 21;27(15):1643-1654. doi: 10.3748/wjg.v27.i15.1643.
- Wang D, Sun S, Zhao Q, et al. Metabolic shifts in tryptophan pathways during acute pancreatitis infections. JCI Insight. 2025 Mar 10;10(5):e186745. doi: 10.1172/jci.insight.186745.
- Badia JM, Amador S, González-Sánchez C, et al. Appropriate Use of Antibiotics in Acute Pancreatitis: A Scoping Review. Antibiotics (Basel). 2024 Sep 18;13(9):894. doi: 10.3390/antibiotics13090894.
- Liu T, Li G. Clinical outcomes and risk factors of invasive candidiasis in patients with infected pancreatic necrosis. Pancreatology. 2025 Nov;25(7):1019-1026. doi: 10.1016/j.pan.2025.09.019.
- Wang L, Zhang W, Dai S, Gao Y, Zhu C, Yu Y. Correlation between the gut microbiota characteristics of hosts with severe acute pancreatitis and secondary intra-abdominal infection. Front Med (Lausanne). 2024 Aug 21;11:1409409. doi: 10.3389/fmed.2024.1409409.
- Frost F, Khaimov V, Senz V, et al. The composition of the stent microbiome is associated with morbidity and adverse events du–ring endoscopic drainage therapy of pancreatic necroses and pseudocysts. Front Med (Lausanne). 2024 Sep 16;11:1462122. doi: 10.3389/fmed.2024.1462122.
- Zou Y, Li N, Li X, et al. Gut microbiota dysbiosis exacerbates acute pancreatitis via Escherichia coli-driven neutrophil heterogeneity and NETosis. Gut Microbes. 2025 Dec 31;18(1):2606480. doi: 10.1080/19490976.2025.2606480.
- Qi-Xiang M, Yang F, Ze-Hua H, et al. Intestinal TLR4 deletion exacerbates acute pancreatitis through gut microbiota dysbiosis and Paneth cells deficiency. Gut Microbes. 2022 Jan-Dec;14(1):2112882. doi: 10.1080/19490976.2022.2112882.
- Jiao J, Liu J, Li Q, et al. Gut Microbiota-Derived Diaminopimelic Acid Promotes the NOD1/RIP2 Signaling Pathway and Plays a Key Role in the Progression of Severe Acute Pancreatitis. Front Cell Infect Microbiol. 2022 Jun 22;12:838340. doi: 10.3389/fcimb.2022.838340.
- Zhang C, Li G, Lu T, et al. The Interaction of Microbiome and Pancreas in Acute Pancreatitis. Biomolecules. 2023 Dec 31;14(1):59. doi: 10.3390/biom14010059.
- Erttmann SF, Swacha P, Aung KM, et al. The gut microbiota prime systemic antiviral immunity via the cGAS-STING-IFN-I axis. Immunity. 2022 May 10;55(5):847-861.e10. doi: 10.1016/j.immuni.2022.04.006.
- Xiao S, Jing S, Jiakui S, et al. Butyrate Ameliorates Intestinal Epithelial Barrier Injury Via Enhancing Foxp3+ Regulatory T-Cell Function in Severe Acute Pancreatitis Model. Turk J Gastroenterol. 2022 Aug;33(8):710-719. doi: 10.5152/tjg.2022.21307.
- Nista EC, Parello S, Brigida M, et al. Exploring the Role of Gut Microbiota and Probiotics in Acute Pancreatitis: A Comprehensive Review. Int J Mol Sci. 2025 Apr 6;26(7):3433. doi: 10.3390/ijms26073433.
- Li J, Pan X, Yang J, et al. Enteral virus depletion modulates experimental acute pancreatitis via toll-like receptor 9 signa–ling. Biochem Pharmacol. 2020 Jan;171:113710. doi: 10.1016/j.bcp.2019.113710.
- Martin-Gallausiaux C, Marinelli L, Blottière HM, Larraufie P, Lapaque N. SCFA: mechanisms and functional importance in the gut. Proc Nutr Soc. 2021 Feb;80(1):37-49. doi: 10.1017/S0029665120006916.
- Pan X, Fang X, Wang F, et al. Butyrate ameliorates caerulein-induced acute pancreatitis and associated intestinal injury by tissue-specific mechanisms. Br J Pharmacol. 2019 Dec;176(23):4446-4461. doi: 10.1111/bph.14806.
- Kim CH. Control of lymphocyte functions by gut microbiota-derived short-chain fatty acids. Cell Mol Immunol. 2021 May;18(5):1161-1171. doi: 10.1038/s41423-020-00625-0.
- Fogelson KA, Dorrestein PC, Zarrinpar A, Knight R. The Gut Microbial Bile Acid Modulation and Its Relevance to Digestive Health and Diseases. Gastroenterology. 2023 Jun;164(7):1069-1085. doi: 10.1053/j.gastro.2023.02.022.
- Tran QT, Tran VH, Sendler M, et al. Role of Bile –Acids and Bile Salts in Acute Pancreatitis: From the Experimental to Clinical Studies. Pancreas. 2021 Jan 1;50(1):3-11. doi: 10.1097/MPA.0000000000001706.
- Jia Y, Shi Y, Wang J, et al. Integrating metagenomics with meta–bolomics for gut microbiota and metabolites profiling in acute pancreatitis. Sci Rep. 2024 Sep 14;14(1):21491. doi: 10.1038/s41598-024-72057-z.
- Zhao M, Cui M, Jiang Q, Wang J, Lu Y. Profile of Pancreatic and Ileal Microbiota in Experimental Acute Pancreatitis. Microorganisms. 2023 Nov 4;11(11):2707. doi: 10.3390/microorga–nisms11112707.
- Wang K, Qin X, Ran T, et al. Causal link between gut microbiota and four types of pancreatitis: a genetic association and bidirectional Mendelian randomization study. Front Microbiol. 2023 Nov 23;14:1290202. doi: 10.3389/fmicb.2023.1290202.
- Zhou R, Wu Q, Yang Z, Cai Y, Wang D, Wu D. The Role of the Gut Microbiome in the Development of Acute Pancreatitis. Int J Mol Sci. 2024 Jan 18;25(2):1159. doi: 10.3390/ijms25021159.
- Zhang C, Chen S, Wang Z, et al. Exploring the mechanism of intestinal bacterial translocation after severe acute pancreatitis: the role of Toll-like receptor 5. Gut Microbes. 2025 Dec;17(1):2489768. doi: 10.1080/19490976.2025.2489768.
- Werge M, Novovic S, Schmidt PN, Gluud LL. Infection increases mortality in necrotizing pancreatitis: A systematic review and meta-analysis. Pancreatology. 2016 Sep-Oct;16(5):698-707. doi: 10.1016/j.pan.2016.07.004.
- van den Berg FF, van Dalen D, Hyoju SK, et al. Western-type diet influences mortality from necrotising pancreatitis and demonstrates a central role for butyrate. Gut. 2021 May;70(5):915-927. doi: 10.1136/gutjnl-2019-320430.
- Besselink MG, van Santvoort HC, Buskens E, et al. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double-blind, placebo-controlled trial. Lancet. 2008 Feb 23;371(9613):651-659. doi: 10.1016/S0140-6736(08)60207-X.