Журнал «Медицина неотложных состояний» 7 (62) 2014
Вернуться к номеру
Needs for the change of surgical clinical classification of acute pancreatitis
Авторы: Riazanov D., Antonevich V., Barannikov K., Potalov S. - State Institute «Zaporozhye Medical Academy of Postgraduate Education of Ministry of Health of Ukraine», Ukraine
Рубрики: Медицина неотложных состояний
Разделы: Клинические исследования
Версия для печати
Статья опубликована на с. 80-83
Abstract
The article generalizes the experience of examination of 886 patients with acute pancreatitis (AP) and its complications. The author offers an original surgical classification of AP and its complications, in which 3 clinical forms of the disease are defined: mild acute pancreatitis, pancreatic necrosis (PN) and pancreatic abscess (PA)), аnd in which such factors are taken into consideration: etiology (non-biliary, biliary), presence of an infection (sterile; infected) with or without a liquid component, the local complications (acute pseudocyst (PC); acute fluid collections (AFC); phlegmon of retroperitoneal space (PhRS)). The classification proposed by us allows working out diagnostic and therapy standards for the use of endovideolaparoscopic treatment, interventional sonography and laparotomy.
Introduction
The most dynamically developing study on AP is determined by sufficiently complete understanding of aetiology and pathogenesis problems based on staging of the course of inflammatory and destructive changes in the gland, by the possibility to determine the state of internal organs and the deepness of necrotic changes of the pancreas tissue and retroperitoneal space with the use of visualizing technologies and standardized programs of intensive conservative therapy of multiple organ dysfunction syndrome (MODS) [2, 6].
However the urge of the surgeons for unification of clinical and instrumental aspects of diagnostics and treatment do not always comply with the single opinion about classification problems of pancreatitis [1, 4].
The number of different classifications of AP approximate to 100. The most successful classification is the international classification by H.G. Beger (1991), which was adopted at the International conciliatory conference in Atlanta, USA in 1992. This classification is an original «starting point» for researchers in their efforts to create more perfect classifications and it objectively deserves comparative with them marks. At the same time this classification doesn’t solve a range of clinical problems and from a surgical point of view it has the following obvious drawbacks [5, 8–10]:
— a long term of clinical diagnosis determination (up to 72 hours);
— absence of an objective clinically significant constant of «sterile pancreatic necrosis» diagnosis determination during the first twenty-four hours of the disease;
— absence in the classification of the wide-spread form of pancreatic infection — of phlegmon of retroperitoneal space;
— absence of consideration of the dynamics and sequence of pancreatic gland (PG) and surrounding tissues morphological transformation during necrotising pancreatitis development;
— absence of consideration of differences in primary localization, interrelation with the ductal system of PG and evolution of PC and AFC;
— absence of fact statement of AFC and pancreatogenic ascite development during necrotising pancreatitis only;
— absence of consideration of possible presence of peripancreatic fluid component during PN;
— the urgent need to work out objective criteria of pre-surgery and intra-surgery differential diagnostics of «infected PN» and of «pancreatic abscess»;
— absence of consideration of the disease etiology and the necessity of the term «acute biliary pancreatitis» (ABP) introduction into the classification.
Pendency of these aspects evolves difficulties not only during consideration and estimation of a clinical form and complications of the disease, but, which is the most important thing, it hampers development and adoption of single international diagnostic and therapeutic standards and protocols. That is why although National standards and protocols of diagnostics and treatment of AP are adopted, they little agree with each other. All this generates excessive variety of interpretations of clinical terms, variants of the disease state and its complications, differences in diagnostic and therapeutic approaches.
The aim of the research
To suggest discussing a unified clinical classification of AP with the aim of standardization of clinical terms, diagnostic and therapeutic tactics and methods of its realization.
Materials and methods
The author have presented the results of examination 886 patients with AP underwent in-patient treatment at the surgery chair with the course of suppurative-septic surgery of the State Institute «Zaporozhye Medical Academy of Postgraduate Education of Ministry of Health of Ukraine» during 1990-2010. Out of those patients there were 361 (40.8 %) with mild AP, 525 (59.2 %) with severe AP. 254 (48.4 %) of patients with severe AP had PN, 29 (5.5 %) of patients had AFC, 95 (18.1 %) of patients had acute PC, 73 (13.9 %) of patients had PA.
Results and their discussion
We present the data of AP patients’ diagnostics and treatment results analysis, which served the basis for the classification modification:
1) The peculiarities of AP activity make the definition of the final diagnosis during the first 72 hours impossible, exclusive of ABP. There is a necessity to repeatedly change the diagnosis during the course of the disease (for example, «severe AP» — «sterile PN» — «AFC» «acute PC»). According to Atlanta classification, every state out of the four mentioned above is an independent clinical form of AP. It is evident that timely definition of a final clinical diagnosis requires concretization of AP clinical forms and its complications with taking into consideration pathomorphological transformation of the disease.
2) Presence of a severe AP during the first 24-hour period should be considered as an objective clinical equivalent of a «sterile PN» diagnosis. Grounds:
a) there are no pathognomonic clinical symptoms, laboratory results, instrumental examination data, estimation scales, which would allow making prognosis of a severe AP transformation into any other local form of AP, which is different from sterile PN;
b) during severe AP sterile PN and/or sterile necrosis of parapancreatic cellular tissue always develop, which is confirmed by the results of autopsy with histological test of tissues of 27 deceased patients with severe AP;
c) during a severe AP there is an average in strength correlation between the massiveness of PN and the frequency of MODS development (r = 0.44): MODS was registered in 486 (92.6 %) patients with severe AP and only in 166 (65.4 %) from 254 patients with PN diagnosed by computer tomography (CT) data. It is evident that singling out patients with PN in the group of patients with severe AP based only on the absence of massive necrotic areas of pancreas during CT is not justified. Establishing identity between the terms «severe AP» and «pancreatic necrosis« will eliminate disagreement and MODS development will be based on the fact of PN regardless of its massiveness;
d) the second infection of necrotic areas is possible in patients with severe AP without local complications and in patients with PN. Its frequency authentically (x2 = 31.5; p < 0.001) differs, making 18 % and 47 %, respectively. The frequency of PC occurance authentically (x2 = 2.5; p > 0.1) doesn’t differ, making up 10.1 % и 16.2 %, respectively.
е) conservative therapy, indications and methods of operative therapy of patients with severe AP without local complications and with sterile PN development are identical;
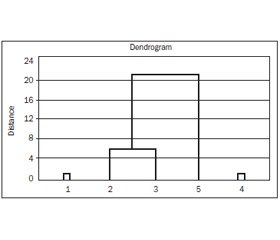
f) mathematical cluster analysis by Ward’s method with the analysis of 28 parameters in patients with AP (etiology; clinical and laboratory signs; severity of the state by Ranson; presence of the syndrome of obstructive jaundice, of system inflammatory response syndrome, MODS, disease complications; second infection of necrotic areas; treatment tactics, autopsy results) and dendrogram construction showed that there exist three clusters. The first cluster is formed by patients with mild AP; the second is formed by severe AP patients without local complications, by patients with sterile PN and AFC, the third is formed by PA patients (fig. 1).
/81/81.jpg)
Hereby, there were no clinical differences determined between the patients with a diagnosis «severe AP» and «sterile PN». Depending on a clinical form of an AP, subdivision of patients into three groups is minimally sufficient.
3) It is determined that 92.3 % of patients with PC and PA had indications of a prior severe AP or a sterile PN in their anamnesis. Besides, localization of PC exclusively in the PG area, frequent determination of a linkage between the PC lacuna with pancreatic duct (84.7 %) allow stating that PC and PA are the result of undergone PN and not independent clinical forms of AP.
4) PC is not a local restricted accumulation of liquid as a result of an unresolved AFC. This is supported by the following facts:
a) differences in the primary localization of PC and AFC: the former are located exclusively in the PG area, the latter are found in other sections of abdominal cavity in 82.8 % of patients. At the same time PC were numerous only in 11 (11.6 %) out of 95 and AFC was in 27 (93.1 %) out of 29 patients; b) differences in evolution of collections: often AFC spontaneously regress during conservative therapy (50 %), but PC didn’t disappear by itself in any of the 6 observed by us cases during the period of up to 5 years; c) differences in the connection with PG duct system: none of the patients with AFC was determined to have suchlike connection; d) the study of PG corrosion preparation while filling the duct system with self-hardening plastic material in deceased patients with necrotising pancreatitis showed that there exists a phenomenon of «depressurization» of the PG duct system, which leads to PC formation [3]; d) high incidence of PC lacuna connection with pancreatic duct (84.7 %).
5) We have determined that AFC was always found in patients with necrotising pancreatitis and was not found in none of the cases in mild AP: a) analysis of the terms of AFC onset in 19 patients showed that already on the second day from the seizure beginning there was AFC onset in 13 (68.4 %) patients, while the medium terms are 9.40 ± 0.45 days and corresponded to the dynamics of PN development; b) 24 (82.7 %) patients with AFC were found necrotic changes in PG during CT, which correspond to stage D in 19 (79.1 %) and E in 5 (20.9 %) of them according to the criteria of Balthazar E.J. and Bradley E.L.; c) 8 patients with mild AP of biliary etiology underwent laparotomic operations and AFC presence was not determined in any of the cases; d) histological study of PG preparations of 3 deceased patients with AFC showed presence of PG necrosis.
Therefore AFC onset in AP patients is a pathognomonic symptom of a necrotising pancreatitis.
6) The necessity of singling out the states with a liquid component in sterile and infected PN is determined by the task of specification of indications for conservatory therapy and use of minimally invasive means of diagnostics and operational therapy, and standardization of the latter. Besides, the use of the term «infected PN with a liquid component» allows reconsidering the term «pancreatic abscess» and differentiating infected PN, PA and PhRS.
The results of combine use of ultrasound diagnostics and CT, surgery, microbiological tests in patients with PN showed that infected PN without a liquid component doesn’t exist. So, while local accumulation of liquid was determined in a non-infected PN in 61.9 % of cases, patients with an infected PN were determined to have local accumulation of liquid in 100 % of cases. At that infected liquid component always has a tendency to increase in dynamics. The determined peculiarity of non-infected PN transformation into an infected PN allowed practically total refusing from the use of a fine-needle aspiration biopsy (FAB) and during PN when the liquid component size is more than 5 cm in diameter performing external drainage of the latter because such a formation is not liable to spontaneous dissipation even without infection. While a small accumulation of liquid when it is technically difficult to perform the drainage, it is advisable to use FAB, which is simpler and less dangerous in these cases. Presence of extended areas of the gland necrosis without a liquid component is not an indication for an FAB.
7) In the classical work by Bittner R. and co-authors [7] morphological, clinical and laboratory criteria for differentiating infected PN and PA were formulated. But the subdivision of the indicated infected forms according to the volume of the liquid part of an empyema, terms of development and morphological characteristics (presence of the capsule during an ultrasound diagnostics and/or CT) is rather conditional, because during an infected PN liquid component (pus) is practically always present, which sometimes exceeds the limits of one anatomical area, while during PA and infected acute PC diffuse PG necroses are frequent. In practice, while performing a surgery on a patient with sequesters of PG and pus, it is difficult to refer them to one of these groups. This is absolutely impossible during the use of close paracentetic drainage methods. At the same time surgery tactics is different. In case of an infected PN a surgery sequesternecrectomy (SNE) is performed. In case of a PA paracentetic drainage is performed and only in case of persisting infection SNE is done. There is a necessity in changing definitions «infected PN» and «PA», in objective differentiation of these clinical states and methods of operational therapy. The following definitions are proposed:
— pancreatic abscess — restricted by a wall accumulation of pus near PG in free abdominal space or in parapancreatic cellular tissue without necrosis of the gland, which is formed over a period of more than 4 weeks from the disease onstart;
— infected PN with a liquid component — infected necrotized areas of PG with local accumulation of pus.
Hereby, two main specific features are determined for the PA diagnosis identification: 1) absence of PG sequestration; 2) localization of the empyema outside of the PG.
It is definite, that such subdivision is conditional and it doesn’t reflect pathogenic mechanisms of development of both states, but it creates real possibilities of unification of diagnosis, treatment plan and methods of treatment, prognostic evaluation of the disease fate.
Thereby, the method of pre-surgical differentiating diagnostics of these states is CT with intravenous contrast enhancement or other similar methods. The method of intra-surgical differentiating diagnostics is results revision: during SNE performing one should consider that the patient has an infected PN with a liquid component with the absence of sequestration of PG — PA.
8) PhRS is not reflected in Atlanta classification. That is why the patients are streamed either to group of an infected PN or PA. Our point of view is that it is possible to define the term «phlegmon of retroperitoneal space» as a diffuse purulent lesion of retroperitoneal cellular tissue which occupies certain anatomic spaces and combines with necrotic changes of PG in 90 % of the cases. On the basis of this definition we held a study with subdivision of patients into groups with PA and PhRS. Analysis of the course of the disease, of general and post-surgery lethality showed that PhRS requires to be singled out into an independent complication of an infected PN, which will allow defining the tactics, methods and terms of operational therapy.
9) We consider that pancreatitis which starts as the result of an obstruction of the distal section of choledoch duct with the development of pancreatic and biliary duct hypertension accompanied by the syndrome of an obstructive jaundice in most cases, should be regarded as an acute biliary pancreatitis.
Suchlike definition of an ABP allows excluding two big categories of patients from this group: 1) patients with an acute cholecystitis and subtend hyperamylasemia; 2) patients with «fermentative cholecystitis». The definition proposed by us proves the need of urgent performing of endoscopic papillosphincterotomy and extraction of concrement, sanitation of the hepaticocholedochus.
The following classification of AP was born as the result of the above mentioned:
1. Mild acute pancreatitis (non-biliary, biliary);
2. Pancreatic necrosis (non-biliary, biliary):
а) sterile:
— without a liquid component;
— with a liquid component.
Complications:
— acute pseudocyst:
- sterile;
- infected;
— acute fluid collections;
в) infected with a liquid component.
Complications: phlegmon of retroperitoneal space;
3. Pancreatic abscess.
The advantages of the proposed AP classification are:
1. It takes into account basic etiological factors of the disease, which are necessary to choose diagnostic and surgery therapeutic tactics.
2. It allows specification of the primary clinical diagnosis in minimum acceptable regulated terms of an urgent situation — up to 24 hours. Exclusion of an ABP, for which diagnosis determination standard is the first two hours from the patient’s admission to hospital.
3. It allows avoiding the necessity of a multiple change of the main final diagnosis. It only adds to it possible local complications of PN in the way of «the main disease complications»
4. It reflects the possibility of emergence of a wide-spread pancreatic infection — PhRS. It makes standardization of its treatment tactics and methods possible.
5. It singles out two clinical states of AP course — absence or presence of PN. This fact is sufficient to determine therapy tactics and treatment, dynamic control both in hospital and after discharging; it allows timely determination and treatment of possible delayed local sequela of PN.
6. It establishes connection between the severity of the patient’s clinical state and morphological changes of PG, retroperitoneal cellular tissue.
7. The thesis about AFC and PC being commemorative signs of a PN informs the doctor of the severity and duration of pancreatitis.
8. It creates conditions for minimally invasive methods at any stage of diagnostics and operational therapy of AP and its complications.
9. It fully reflects clinical forms of AP and local organ complications on the part of PG, which allows standardization of diagnostic, conservative and operational therapy, creation of precise and clear algorithms of patient’s management at any stage of pancreatitis clinical course and its complications and also defining acceptable lethality rate.
Conclusion
The clinical classification proposed by us allows working out diagnostic and therapy standards for the use of endovideolaparoscopic treatment, interventional sonography, laparotomy, that should be staging and directed, first of all, to lowing general and post-operational lethality, which in our opinion, shouldn’t exceed 2 % and 5 % accordingly.
It is potentially productive to create a work group on developing unified international surgery diagnostic and therapy standards of management of AP patients.
1. Березницький Я.С., Яльченко Н.А., Кабак Г.Г., Кутовий М.О. Класифікаційні ознаки гострого панкреатиту як основа клінічного діагнозу і цілеспрямованої адекватної медичної допомоги // Экспериментальная и клиническая медицина. — 2004. — № 3. — С. 16-19.
2. Вашетко Р.В., Толстой А.Д., Курыгин А.А., Стойко Ю.М. Острый панкреатит и травмы поджелудочной железы. — СПб.: Питер, 2000. — 320 с.
3. Даценко Б.М., Тамм Т.И., Крамаренко К.А. Острый панкреатит. Учебное пособие для врачей. Харьковская медицинская академия последипломного образования. — Х.: Прапор, 2004. — 88 с.
4. Коновалов Є.П. та співавт. Стандарти діагностики і лікування гострого панкреатиту. — Київ, 2004. — 27 с.
5. Филимонов М.И., Гельфанд Б.Р., Бурневич С.З., Орлов Б.Б., Цыденжапов Е.Ц. Острый панкреатит: Пособие для врачей / Под редакцией В.С. Савельева. — М.: Издательство НЦССХ им. А.Н. Бакулева РАМН, 2000. — 60 с.
6. Яицкий Н.А., Седов В.М., Сопия Р.А. Острый панкреатит. — М.: МЕДпресс-информ, 2003. — 224 с.
7. Bittner R., Block S., Buchler M., Beger H.G. Pancreatitis abscess and infected pankreonekrosis. Different local septic complications of acute pancreatitis // Dig. Dis. Sci. — 1987. — Vol. 32(10). — P. 1082-1087.
8. Bradley E.L. A clinical based classification system for acute pancreatitis // Arch. Surg. — 1993. — Vol. 128. — P. 586-589.
9. Folsch U.R., Nitsche R., Ludtke R. et al. Early ERCP and papillotomy compared with conservative management for acute biliary pancreatitis // The German Study Group on Acute Pancreatitis // Engl. J. Med. — 1997. — Vol. 336. — P. 237-242.
10. Toh S.K., Phillips S., Johnson C.D. A prospective audit against national standards of the presentation and management of acute pancreatitis in the South of England // Gut. — 2000. — Vol. 46. — P. 239-243.