Статья опубликована на с. 9-14, 15
В последнее время возобновился интерес к вопросу о роли симпатического отдела нервной системы при артериальной гипертензии. Причинами тому послужили: 1) результаты исследований, показывающие, что нарушения со стороны симпатического отдела нервной системы могут оказывать влияние на развитие и прогрессирование поражений в органах-мишенях; 2) разработки новых терапевтических подходов, направленных на нормализацию артериального давления; 3) данные о том, что активация симпатической нервной системы оказывает негативное прогностическое влияние на заболеваемость и смертность от различных сердечно-сосудистых заболеваний. Это подчеркивает важность модуляции симпатической активации в качестве целевого показателя немедикаментозных методов, а также фармакотерапии, направленной на снижение высоких цифр артериального давления. Тем не менее по-прежнему имеется недостаточное количество данных. Для определения роли генетических факторов, отношения между функцией симпатической нервной системы и поражениями органов-мишеней, а также влияния комбинированной фармакотерапии на функции симпатической нервной системы и ее корреляции с мероприятиями по контролю артериального давления необходимо провести дальнейшие исследования.
Предпосылки
Возобновление интереса со стороны исследователей и клиницистов к роли симпатической нервной системы при артериальной гипертензии и ее актуальность в качестве мишени немедикаментозных методов и фармакологической терапии основывается на целом ряде причин. Во-первых, результаты последних исследований показывают, что нарушения симпатической иннервации приводят к развитию и прогрессированию поражения органов-мишеней, независимо от повышения артериального давления [1, 2]. Во-вторых, разработаны новые терапевтические подходы для лечения и контроля высокого артериального давления в условиях высокого риска его развития, например при резистентной артериальной гипертензии, а именно: стимуляция каротидных барорецепторов и радиочастотная абляция почечного нерва [2]. И наконец, имеются данные о том, что при целом ряде основных сердечно-сосудистых заболеваний, таких как застойная сердечная недостаточность, инсульт, инфаркт миокарда и почечная недостаточность, которые патогенетически связаны с развитием артериальной гипертензии, активация симпатической нервной системы имеет независимое неблагоприятное прогностическое значение в плане заболеваемости и смертности [3–7].
В совокупности эти данные подчеркивают важность модулирования активации симпатической нервной системы с целью как немедикаментозной, так и медикаментозной терапии, направленной на снижение высоких цифр артериального давления.
Активация симпатической нервной системы при артериальной гипертензии
Состояния, при которых развивается эссенциальная гипертензия, как было доказано, характеризуются не только нарушением тонуса парасимпатической, но и выраженным гипертонусом симпатической нервной системы, что проявляется повышением частоты сердечных сокращений в покое [1]. Симпатическая активация приводит к развитию этих гемодинамических изменений вследствие своих известных положительных хронотропных эффектов, которые свойственны главному адренергическому нейромедиатору норадреналину [1]. На раннем этапе развития гипертонического криза, при пограничной артериальной гипертензии, вероятнее всего, задействованы оба этих нейрогенных нарушения [2]. Тем не менее, если выраженность вагусной дисфункции при клинических состояниях, характеризующихся более тяжелым увеличением артериального давления, остается стабильной, то активация симпатической нервной системы постепенно усиливается, что вызывает более тяжелую степень гипертензивных состояний [8]. Это было доказано, в частности, с помощью прямых методов исследования функций симпатической нервной системы человека, таких как клиническая микронейрография, которая, непосредственно с помощью записи эфферентного постганглионарного симпатического нейронного разряда в малоберцовом или плечевом нерве человека, позволяет преодолевать известные ограничения анализа плазменного адренергического маркера норадреналина [2].
Вышеупомянутая симпатическая дисрегуляция была продемонстрирована на различных стадиях гипертонической болезни (легкой, умеренной и тяжелой), при артериальной гипертензии у пациентов молодого, среднего и пожилого возраста, артериальной гипертензии «белого халата», латентной гипертензии и артериальной гипертензии, вызванной беременностью [1, 2]. Недавно были задокументированы и другие клинические состояния, которые ассоциировались с гиперактивностью симпатического отдела нервной системы. Они включают в себя гипертензию во время сна и бодрствования, гипертензию, осложненную синдромом апноэ сна, метаболическим синдромом или почечной недостаточностью, и истинную резистентную гипертензию [1, 2, 9]. Наконец, следует отметить, что: 1) вторичные формы артериальной гипертензии, такие как реноваскулярная гипертензия, вероятно, не связаны с симпатической активацией; 2) механизмы, ответственные за развитие артериальной гипертензии, обусловленной адренергическим перенапряжением, возможно, являются комплексными, включая изменения в нейрогенной, рефлекторной, а также метаболической модуляции тонуса симпатической нервной системы [1, 2].
Клиническая значимость артериальной гипертензии, связанной с симпатической гиперактивностью
В настоящее время известны прямые и косвенные доказательства того, что состояния, связанные с симпатической активацией, приводят к развитию сердечных и сосудистых изменений, тем самым вызывая повышение заболеваемости и смертности в случаях нелеченой артериальной гипертензии [1, 2]. Что касается изменений со стороны сердца, есть доказательства того, что повышенный тонус симпатической нервной системы, воздействующей на сердце, определяется у больных артериальной гипертензией с гипертрофией левого желудочка или даже с диастолической дисфункцией левого желудочка; это подтверждает концепцию о том, что кроме уровня артериального давления крайне важную роль в определении структурных и функциональных изменений миокарда, которые имеют место при различных клинических гипертонических состояниях, играют и другие факторы [10, 11]. Также было доказано, что симпатическая активация участвует в развитии и прогрессировании ремоделирования сосудов, эндотелиальной дисфункции, а также в повышении артериального тонуса, которые обусловливают развитие гипертонических состояний [12, 13]. Наконец, в исследованиях последних лет доказано, что как метаболические нарушения, так и заболевания почек, проявляющиеся артериальной гипертензией не только на поздних, но и на более ранних стадиях, действительно связаны с изменениями в симпатической нервной системе, которые, вероятно, потенцируют адренергическую перегрузку, встречающуюся уже при неосложненной артериальной гипертензии [14, 15].
Торможение активности симпатического отдела нервной системы как мишень гипотензивной терапии
Как показано в табл. 1, уменьшение симпатического тонуса, воздействующего на сердечно-сосудистую систему, может вызывать целый ряд благоприятных сердечно-сосудистых и кардиометаболических эффектов. Самыми актуальными из них с клинической точки зрения являются: 1) равномерный контроль артериального давления в течение суток; 2) уменьшение диапазона колебания артериального давления в течение суток, 3) регрессирование поражений органов-мишеней; 4) улучшение метаболических нарушений, связанных с гипертонией. С другой стороны, симпатомиметики оказывают совершенно противоположное действие на сердечно-сосудистую и кардиометаболическую системы (табл. 1); они прямо или косвенно: 1) уменьшают равномерный контроль артериального давления в течение суток; 2) повышают диапазон колебания артериального давления в течение суток; 3) нивелируют регрессирование сердечных поражений; 4) ухудшают метаболические нарушения, связанные с гипертонией.
/12.jpg)
Симпатические эффекты немедикаментозной и медикаментозной терапии
Что касается немедикаментозных методов, имеются неопровержимые доказательства воздействия на симпатическую систему низкокалорийной диеты и программы выполнения регулярных физических упражнений [16, 17]. Поскольку обе процедуры оказывают четко выраженное влияние в плане снижения артериального давления, величина которого часто связана со степенью симпатической блокады, была выдвинута гипотеза о том, что действие этих двух антигипертензивных методов обусловлено их симпатоблокирующими эффектами [16, 17]. И наоборот, длительная диета с низким содержанием натрия, как сообщалось, приводит к увеличению уже повышенного адренергического тонуса [16, 17]. Это, вероятно, связано с тем, что диета с низким содержанием натрия вызывает гиперинсулинемию и активирует ренин-ангиотензиновую систему, т.е. два эффекта, которые способствуют возбуждению симпатической нервной системы и оказывают влияние на барорефлекторный контроль со стороны как вагального, так и симпатического тонуса [1, 2]. Недавно были успешно разработаны две инвазивные процедуры: имплантация устройства, способного стимулировать каротидные барорецепторы (а следовательно, ингибировать активность симпатической нервной системы и повышать барорефлекторный контроль вагального тонуса, воздействующего на сердце), и денервация почечного симпатического нерва через катетер, размещенный в почечной артерии и подключенный к радиочастотному генератору. Обе эти процедуры находятся на стадии клинических исследований с целью изучения их гипотензивного эффекта, который, вероятно, связан с уменьшением симпатического тонуса [18, 19].
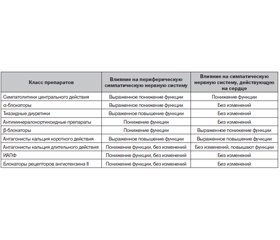
Что касается влияния гипотензивных препаратов на сердечно-сосудистую вегетативную функцию, имеются доказательства того, что, как показано в табл. 2, некоторые фармакологические классы антигипертензивных препаратов (например, бета-блокаторы, ингибиторы ангиотензинпревращающего фермента (ИАПФ) и блокаторы рецепторов ангиотензина II) могут оказывать выраженное симпатоблокирующее действие, в то время как другие классы вообще его не оказывают (антагонисты кальция длительного действия) или даже усиливают адренергический тонус в отношении сердечно-сосудистой системы (диуретики, антагонисты кальция короткого действия) [16, 20–22].
/13.jpg)
В настоящее время имеется недостаточное количество информации о действиях различных комбинаций антигипертензивных препаратов на функцию вегетативной нервной системы, воздействующей на сердечно-сосудистую систему, и вся она основывается на косвенных и, таким образом, менее чувствительных маркерах тонуса симпатической нервной системы, таких как плазменная концентрация норадреналина.
Выводы
Еще предстоит определить целый ряд вопросов, связанных с ролью симпатической нервной системы при артериальной гипертензии:
— роль генетических факторов в перегрузке симпатической нервной системы, обусловленной гипертензией;
— отношения между различными показателями функции симпатической нервной системы и новыми маркерами повреждения органов-мишеней/артериальной дисфункции;
— воздействие комбинированной лекарственной терапии на функции симпатической нервной системы и ее возможная связь с контролем артериального давления.
Комментарий специалиста
Статья Guido Grassi (университет Milano-Bicocca, Милан) вызвала интерес редакции в связи с ренессансом роли доминирования гиперактивности симпатической системы в развитии и прогрессировании заболеваний почек. Акцент на состоянии симпатической системы тем более интересен, что автор является сотрудником ведущего университета (клиническая база госпиталя г. Монза). Не останавливаясь на вопросах патогенеза, хотелось бы еще раз вернуться к современной терапии.
Как следует из табл. 2 цитируемой статьи, лишь блокаторы РААС (ИАПФ/БРА и не приведенный в таблице прямой ингибитор ренина, а также блокаторы минералокортикоидных рецепторов) понижают функцию симпатической системы, негативно не влияя на сердце. Эти препараты принято называть ренопротекторными, так как с доказательной точки зрения лишь они способствуют увеличению сроков до начала почечнозаместительной терапии при ХБП.
Вместе с тем существуют по крайней мере два фактора, которые заставляют расширять спектр назначаемых препаратов, снижающих гиперактивность симпатической системы. Первый — это необходимость комбинированной терапии артериальной гипертензии при ХБП, которая возрастает по мере снижения СКФ. Второй — высокая вероятность необходимости отмены блокаторов РААС при ХБП 4–5-й стадий в связи с их способностью снижать СКФ.
В связи с этим возникает вопрос: что использовать в дополнение или чем заменить блокаторы РААС при прогрессирующей утрате функций почек? Ответ опять-таки в табл. 2 цитируемого автора: симпатолитики, бета-блокаторы (практически только небиволол из-за его свойства снижать центральное АД) и блокаторы кальциевых каналов длительного действия (а правильнее — низковольтных рецепторов), из которых в Украине в наличии только лерканидипин. О современных симпатолитиках мы пишем практически в каждом номере журнала: это блокатор имидазолиновых рецепторов — Моксогамма (0,2, 0,3 и 0,4 мг моксонидина).
Таким образом, наличие и формирование доказательной базы позволяют рекомендовать формулу использования блокаторов гиперактивности симпатической системы, в той или иной мере обладающих ренопротекторным эффектом: БРИМОНЕЛ — БР(А) И(АПФ) МО(ксонидин) НЕ(биволол) Л(ерканидипин). Их комбинация не рекомендуется для широкой практики и с осторожностью применяется в нефрологии при СКФ более 60 мл/мин. Комбинация ИАПФ и БРА, очевидно, на сегодняшний день является: 1) патогенетически обоснованной; 2) клинически эффективной и 3) достоверно снижающей смертность при ХБП.
Список литературы
1. Mancia G., Grassi G. The autonomic nervous system and hypertension // Circ. Res. 2014, May 23; 114(11): 1804-14.
2. Grassi G., Mark A., Esler M. The sympathetic nervous system alterations in human hypertension // Circ. Res. 2015, Mar 13; 116(6): 976-90.
3. Cohn J.N., Levine T.B., Olivari M.T., Garberg V., Lura D., Francis G.S., Simon A.B., Rector T. Plasma norepinephrine as a guide to prognosis in patients with chronic congestive heart failure // N. Engl. J. Med. 1984, Sep 27; 311(13): 819-23.
4. Brunner-La Rocca H.P., Esler M.D., Jennings G.L., Kaye D.M. Effect of cardiac sympathetic nervous activity on mode of death in congestive heart failure // Eur. Heart J. 2001 Jul; 22(13): 1136-43.
5. Sander D., Winbeck K., Klingelhöfer J., Etgen T., Conrad B. Prognostic relevance of pathological sympathetic activation after acute thromboembolic stroke // Neurology. 2001, Sep 11; 57(5): 833-8.
6. Zoccali C., Mallamaci F., Parlongo S., Cutrupi S., Benedetto F.A., Tripepi G., Bonanno G., Rapisarda F., Fatuzzo P., Seminara G., Cataliotti A., Stancanelli B., Malatino L.S. Plasma norepinephrine predicts survival and incident cardiovascular events in patients with end-stage renal disease // Circulation. 2002, Mar 19; 105(11): 1354-9.
7. Barretto A.C., Santos A.C., Munhoz R., Rondon M.U., Franco F.G., Trombetta I.C., Roveda F., de Matos L.N., Braga A.M., Middlekauff H.R., Negrão C.E. Increased muscle sympathetic nerve activity predicts mortality in heart failure patients // Int. J. Cardiol. 2009, Jul 10; 135(3): 302-7.
8. Grassi G., Cattaneo B.M., Seravalle G., Lanfranchi A., Mancia G. Baroreflex control of sympathetic nerve activity in essential and secondary hypertension // Hypertension. 1998 Jan; 31(1): 68-72.
9. Grassi G., Seravalle G., Brambilla G., Pini C., Alimento M., Facchetti R., Spaziani D., Cuspidi C., Mancia G. Marked sympathetic activation and baroreflex dysfunction in true resistant hypertension // Int. J. Cardiol. 2014, Dec 20; 177(3): 1020-5.
10. Burns J., Sivananthan M.L., Ball S.G., Mackintosh A.E., Mary D.A., Greenwood J.P. Relationship between central sympathetic drive and magnetic resonance determined left ventricular mass in essential hypertension // Circulation. 2007, Apr 17; 115(15): 1999-2005.
11. Grassi G., Seravalle G., Quarti-Trevano F., Dell’Oro R., Arenare F., Spaziani D., Mancia G. Sympathetic and baroreflex cardiovascular control in hypertension-related left ventricualr diastolic dysfunction // Hypertension. 2009 Feb; 53(2): 205-9.
12. Laurent S., Safar M.E. Large artery damage: measurement and clinical importance // Mancia G., Grassi G., Redon J. (eds). Manual of Hypertension of the European Society of Hypertension. CRC Press (London), 2014: p191-201.
13. Heagerty A.M., Withers S.B., Izzard A.S., Greenstein A.S., Aghamohammadzadeh R. Small artery structure and function in hypertension // Mancia G., Grassi G., Redon J. (eds). Manual of Hypertension of the European Society of Hypertension. CRC Press (London), 2014: 203-210.
14. Esler M., Straznicky N., Eikelis N., Masuo K., Lambert G., Lambert E. Mechanisms of sympathetic activation in obesity-related hypertension // Hypertension. 2006 Nov; 48(5): 787-96.
15. Grassi G., Quarti-Trevano F., Seravalle G., Arenare F., Volpe M., Furiani S., Dell’Oro R., Mancia G. Early sympathetic activation in the initial clinical stages of chronic renal failure // Hypertension. 2011 Apr; 57(4): 846-51.
16. Grassi G. Counteracting the sympathetic nervous sytem in hypertension // Curr. Opin. Nephrol. Hypertens. 2004 Sep; 13(5): 513-9.
17. Mancia G., Fagard R., Narkiewicz K., Redón J., Zanchetti A., Böhm M., Christiaens T., Cifkova R., De Backer G., Dominiczak A., Galderisi M., Grobbee D.E., Jaarsma T., Kirchhof P., Kjeldsen S.E., Laurent S., Manolis A.J., Nilsson P.M., Ruilope L.M., Schmieder R.E., Sirnes P.A., Sleight P., Viigimaa M., Waeber B., Zannad F.; Task Force Members. 2013 ESH/ESC Guidelines for the management of arterial hypertension // J. Hypertens. 2013 Jul; 31(7): 1281-357.
18. Bisognano J.D., Bakris G., Nadim M.K., Sanchez L., Kroon A.A., Schafer J., de Leeuw P.W., Sica D.A. Baroreflex activation therapy lowers blood pressure in patients with resistant hypertension // J. Am. Coll. Cardiol. 2011, Aug 9; 58(7): 765-73.
19. Grassi G., Seravalle G., Brambilla G., Trabattoni D., Cuspidi C., Corso R., Pieruzzi F., Genovesi S., Stella A., Facchetti R., Spaziani D., Bartorelli A., Mancia G. Blood pressure responses to renal denervation precede and are independent of the sympathetic and baroreflex effects // Hypertension. 2015 Jun; 65(6): 1209-16.
20. Sica D. Centrally acting antihypertensive agents: an update // J. Clin. Hypertens. (Greenwich). 2007 May; 9(5): 399-405.
21. Wray D.W., Supiano M.A. Impact of aldosterone receptor blockade compared with thiazide therapy on sympathetic nervous system function in geriatric hypertension // Hypertension. 2010 May; 55(5): 1217-23.
22. Neumann J., Ligtenberg G., Oey L., Koomans H.A., Blankestjin P. Moxonidine normalizes sympathetic hyperactivity in patients with eprosartan-treated chronic renal failure // J. Am. Soc. Nephrol. 2004 Nov; 15(11): 2902-7.
Согласие автора на публикацию в журнале «Почки» получено 24.12.15
Перевод с англ. П. Огилько
Оригинал статьи опубликован в e-journal of the ESC Council for Cardiology Practice, 24 Nov 2015, Vol. 13, № 36
/15.jpg)

/12.jpg)
/13.jpg)