Журнал «Медицина неотложных состояний» Том 16, №8, 2020
Вернуться к номеру
Вітамін С при критичних станах: від експерименту до клініки (частина 1)
Авторы: Чуклін С.М., Чуклін С.С.
Львівська обласна клінічна лікарня, м. Львів, Україна
Рубрики: Медицина неотложных состояний
Разделы: Справочник специалиста
Версия для печати
Вітамін С (аскорбінова кислота) відіграє важливу фізіологічну роль у численних метаболічних функціях. Він також є кофактором у синтезі важливих речовин, зокрема катехоламінів і вазопресину. Зниження рівня аскорбінової кислоти відзначено при різних захворюваннях і часто супроводжує тяжкий стан хворого. Метою цієї статті є огляд сучасних уявлень щодо фізіологічної ролі вітаміну С та експериментального обґрунтування його застосування при критичних станах у хірургічних хворих. Розглядається фармакодинаміка аскорбінової кислоти, можливість застосування високих доз вітаміну С. Для пошуку літературних джерел використовувалася база MEDLINE на платформі PubMed за ключовими словами: вітамін С, сепсис, шок, травма, опіки.
Витамин С (аскорбиновая кислота) играет важную физиологическую роль в многочисленных метаболических функциях. Он также является кофактором в синтезе важных веществ, в частности катехоламинов и вазопрессина. Снижение уровня аскорбиновой кислоты отмечено при различных заболеваниях и часто сопровождает тяжелое состояние больного. Целью этой статьи является обзор современных представлений о физиологической роли витамина С и экспериментального обоснования его применения при критических состояниях у хирургических больных. Рассматривается также фармакодинамика аскорбиновой кислоты, возможность применения высоких доз витамина С. Для поиска литературных источников использовалась база MEDLINE на платформе PubMed по ключевым словам: витамин С, сепсис, шок, травма, ожоги.
Vitamin C (ascorbic acid) plays an important physiological role in numerous metabolic functions. It is also a cofactor in the synthesis of important substances, in particular catecholamines and vasopressin. A decrease in the level of ascorbic acid has been noted in various diseases, often accompanying the severity of the patient’s state. The aim of this article is to review the current knowledge on the physiological role of vitamin C and the experimental evidences of its use in critically ill surgical patients. The pharmacodynamics of ascorbic acid, the possibility of using high doses of vitamin C are also considered. MEDLINE database on the PubMed platform was used to search for the literature sources with key words: vitamin C, sepsis, shock, trauma, burns.
вітамін С; фармакокінетика; фізіологічна роль; сепсис; шок; травма; опіки
витамин С; фармакокинетика; физиологическая роль; сепсис; шок; травма; ожоги
vitamin C; pharmacokinetics; physiological role; sepsis; shock; trauma; burns
Вступ
Фармакокінетика
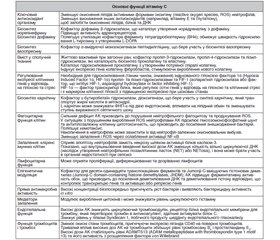
Функції вітаміну С
Застосування вітаміну С в експерименті
Висновки
- Drouin G., Godin J.-R., Pagé B. The genetics of vitamin C loss in vertebrates. Curr. Genom. 2011. Vol. 12. P. 371-378.
- Lachapelle M.Y., Drouin G. Inactivation dates of the human and guinea pig vitamin C genes. Genetica. 2011. Vol. 139. P. 199-207.
- Pauling L. Evolution and the need for ascorbic acid. Proc. Natl. Acad. Sci. USA. 1970. Vol. 67. P. 1643-1648.
- May J.M., Harrison F.E. Role of vitamin C in the function of the vascular endothelium. Antioxid. Redox Signal. 2013. Vol. 19. P. 2068-2083.
- Granger M., Eck P. Dietary vitamin C in human health. Adv. Food Nutr. Res. 2018. Vol. 83. P. 281-310.
- Daruwala R., Song J., Koh W.S. et al. Cloning and functional characterization of the human sodium-dependent vitamin C transporters hSVCT1 and hSVCT2. FEBS Lett. 1999. Vol. 460. P. 480-484.
- Tsukaguchi H., Tokui, T., Mackenzie B. et al. A family of mammalian Na+-dependent L-ascorbic acid transporters. Nature. 1999. Vol. 399. P. 70-75.
- Corpe C.P., Eck P., Wang J. et al. Intestinal dehydroascorbic acid (DHA) transport mediated by the facilitative sugar transporters, GLUT2 and GLUT8. J. Biol. Chem. 2013. Vol. 288. P. 9092-9101.
- Kuhn V., Diederich L., Keller T.C.S. et al. Red blood cell function and dysfunction: Redox regulation, nitric oxide metabolism, anemia. Antioxid. Redox Signal. 2017. Vol. 26. P. 718-742.
- Berger M.M., Oudemans-van Straaten H.M. Vitamin C supplementation in the critically ill patient. Curr. Opin. Clin. Nutr. Metab. Care. 2015. Vol. 18. P. 193-201.
- Savini I., Rossi A., Pierro C. et al. SVCT1 and SVCT2: key proteins for vitamin C uptake. Amino Acids. 2008. Vol. 34. P. 347-355.
- Levine M., Conry-Cantilena C., Wang Y. et al. Vitamin C pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc. Natl. Acad. Sci. USA. 1996. Vol. 93. P. 3704-3709.
- Padayatty S.J., Sun H., Wang Y. et al. Vitamin C pharmacokinetics: Implications for oral and intravenous use. Ann. Intern. Med. 2004. Vol. 140. P. 533-537.
- Hager D.N., Martin G.S., Sevransky J.E., Hooper M.H. Glucometry when using vitamin C in sepsis: A note of caution. Chest. 2018. Vol. 154. P. 228-229.
- Cho J., Ahn S., Yim J. et al. Influence of vitamin C and maltose on the accuracy of three models of glucose meters. Ann. Lab. Med. 2016. Vol. 36(3). P. 271-274.
- Meng Q.H., Irwin W.C., Fesser J., Massey K.L. Interference of ascorbic acid with chemical analytes. Ann. Clin. Biochem. 2005. Vol. 42 (Pt 6). P. 475-477.
- Smith K.E., Brown C.S., Manning B.M. et al. Accuracy of Point of Care Blood Glucose Level Measurements in Critically Ill Patients with Sepsis Receiving High-Dose Intravenous Vitamin C. Pharmacotherapy. 2018. Vol. 38. P. 1155-1161.
- Moskowitz A., Andersen L.W., Huang D.T. et al. Ascorbic acid, corticosteroids, and thiamine in sepsis: a review of the biologic rationale and the present state of clinical evaluation. Crit. Care. 2018. Vol. 22. P. 283.
- Lowes D.A., Webster N.R., Galley H.F. Dehydroascorbic acid as pre-conditioner: Protection from lipopolysaccharide induced mitochondrial damage. Free Radic. Res. 2010. Vol. 44. P. 283-292.
- Yimcharoen M., Kittikunnathum S., Suknikorn C. et al. Effects of ascorbic acid supplementation on oxidative stress markers in healthy women following a single bout of exercise. J. Int. Soc. Sports Nutr. 2019. Vol. 16. P. 2.
- Marik P.E. Hydrocortisone, Ascorbic Acid and Thiamine (HAT therapy) for the treatment of sepsis. Focus on ascorbic acid. Nutrients. 2018. Vol. 10. P. 1762.
- Marik P.E. Vitamin C for the treatment of sepsis: The scientific rationale. Pharmacol. Ther. 2018. Vol. 189. P. 63-70.
- Carr A.C., Maggini S. Vitamin C and Immune Function. Nutrients. 2017. Vol. 9. P. 1211.
- Padayatty S.J., Levine M. Vitamin C physiology: the known and the unknown and Goldilocks. Oral. Dis. 2016. Vol. 22. P. 463-493.
- Van Gorkom G.N.Y., Klein Wolterink R.G.J., Van Elssen C.H.M.J. et al. Influence of Vitamin C on Lymphocytes: An Overview. Antioxidants. 2018. Vol. 7. E41.
- Tanaka M., Muto N., Gohda E., Yamamoto I. Enhancement by ascorbic acid 2-glucoside or repeated additions of ascorbate of mitogen-induced IgM and IgG productions by human peripheral blood lymphocytes. Jpn. J. Pharmacol. 1994. Vol. 66. P. 451-456.
- Gao Y.L., Lu B., Zhai J.H. et al. The parenteral vitamin C improves sepsis and sepsis-induced multiple organ dysfunction syndrome via preventing cellular immunosuppression. Mediators Inflamm. 2017. Vol. 2017. P. 4024672.
- Nusgens B.V., Humbert P., Rougier A. et al. Stimulation of collagen biosynthesis by topically applied vitamin C. Eur. J. Dermatol. 2002. Vol. P. 12. XXXII-XXXIV.
- Teuwen L.A., Draoui N., Dubois C., Carmeliet P. Endothelial cell metabolism: an update anno 2017. Curr. Opin. Hematol. 2017. Vol. 24. P. 240-247.
- Wang Z.Y., Liu Y.Y., Liu G.H. et al. l-Carnitine and heart disease. Life Sci. 2018. Vol. 194. P. 88-97.
- Puskarich M.A., Kline J.A., Krabill V. et al. Preliminary safety and efficacy of L-carnitine infusion for the treatment of vasopressor-dependent septic shock: A randomized control trial. JPEN. 2014. Vol. 38. P. 736-743.
- Oudemans-van Straaten H.M., Spoelstra-de Man A., de Waard M.C. Vitamin C revisited. Crit. Care. 2014. Vol. 18. P. 460.
- Carr A.C., Shaw G., Fowler A.A., Natarajan R. Ascorbate-dependent vasopressor synthesis — a rationale for vitamin C administration in severe sepsis and septic shock? Crit. Care. 2015. Vol. 19. P. 418.
- Bornstein S.R., Yoshida-Hiroi M., Sotiriou S. et al. Impaired adrenal catecholamine system function in mice with deficiency of the ascorbic acid transporter (SVCT2). FASEB J. 2003. Vol. 17. P. 1928-1930.
- Dillon P.F., Root-Bernstein R., Robinson N.E. et al. Receptor-mediated enhancement of beta adrenergic drug activity by ascorbate in vitro and in vivo. PloS One. 2010. Vol. 5. e15130.
- May J.M., Qu Z.C., Meredith M.E. Mechanisms of ascorbic acid stimulation of norepinephrine synthesis in neuronal cells. Biochem. Biophys. Res. Commun. 2012. Vol. 426. P. 148-152.
- Wilson J.X. Evaluation of vitamin C for adjuvant sepsis therapy. Antioxid. Redox Signal. 2013. Vol. 19. P. 2129-2140.
- Kim S.R., Kim Y.M., Park E.J. et al. Ascorbic acid reduces HMGB1 secretion in lipopolysaccharide-activated RAW 264.7 cells and improves survival rate in septic mice by activation of Nrf2/HO-1 signals. Biochem. Pharmacol. 2015. Vol. 95. P. 279-289.
- Hagel A.F., Layritz C.M., Hagel W.H. et al. Intravenous infusion of ascorbic acid decreases serum histamine concentrations in patients with allergic and non-allergic diseases. Naunyn-Schmiedebergs Arch. Pharmacol. 2013. Vol. 386. P. 789-793.
- Hattori M., Yamazaki M., Ohashi W. et al. Critical role of endogenous histamine in promoting end-organ tissue injury in sepsis. Intensive Care Med. Exp. 2016. Vol. 4. P. 36.
- Kashiouris M.G., L’Heureux M., Cable C.A. et al. The Emerging Role of Vitamin C as a Treatment for Sepsis. Nutrients. 2020. Vol. 12. P. 292.
- Langlois P.L., Lamontagne F.L. Vitamin C for the critically ill: Is the evidence strong enough? Nutrition. 2019. Vol. 60. P. 185-190.
- Carr A., Frei B. Does vitamin C act as a pro-oxidant under physiological conditions? FASEB J. 1999. Vol. 13. P. 1007-1024.
- Larsen R., Gozzelino R., Jeney V. et al. A central role for free heme in the pathogenesis of severe sepsis. Sci. Transl. Med. 2010. Vol. 2. 51ra71.
- Seo M.Y., Lee S.M. Protective effect of low dose of ascorbic acid on hepatobiliary function in hepatic ischemia/reperfusion in rats. J. Hepatol. 2002. Vol. 36. P. 72-77.
- Park S.W., Lee S.M. Antioxidant and prooxidant properties of ascorbic acid on hepatic dysfunction induced by cold ischemia/reperfusion. Eur. J. Pharmacol. 2008. Vol. 580. P. 401-406.
- Manzella J.P., Roberts N.J. Human macrophage and lymphocyte responses to mitogen stimulation after exposure to influenza virus, ascorbic acid, and hyperthermia. J. Immunol. 1979. Vol. 123. P. 1940-1944.
- Siegel B.V. Enhancement of interferon production by poly(rI)-poly(rC) in mouse cell cultures by ascorbic acid. Nature. 1975. Vol. 254. P. 531-532.
- Motl J., Radhakrishnan J., Ayoub I.M. et al. Vitamin C compromises cardiac resuscitability in a rat model of ventricular fibrillation. Am. J. Ther. 2014. Vol. 21. P. 352-357.
- Levine M., Padayatty S.J., Espey M.G. Vitamin C: A concentration-function approach yields pharmacology and therapeutic discoveries. Adv. Nutr. 2011. Vol. 2. P. 78-88.
- Muhlhofer A., Mrosek S., Schlegel B. et al. High-dose intravenous vitamin C is not associated with an increase of pro-oxidative biomarkers. Eur. J. Clin. Nutr. 2004. Vol. 58. P. 1151-1158.
- Dennis J.M., Witting P.K. Protective Role for Antioxidants in Acute Kidney Disease. Nutrients. 2017. Vol. 9. E718.
- Armour J., Tyml K., Lidington D., Wilson J.X. Ascorbate prevents microvascular dysfunction in the skeletal muscle of the septic rat. J. Appl. Physiol. 2001. Vol. 90. P. 795-803.
- Tyml K., Li F., Wilson J.X. Delayed ascorbate bolus protects against maldistribution of microvascular blood flow in septic rat skeletal muscle. Crit. Care Med. 2005. Vol. 33. P. 1823-1828.
- Fisher B.J., Seropian I.M., Kraskauskas D. et al. Ascorbic acid attenuates lipopolysaccharide-induced acute lung injury. Crit. Care Med. 2011. Vol. 39. P. 1454-1460.
- Fisher B.-J., Kraskauskas D., Martin E.J. et al. Mechanisms of attenuation of abdominal sepsis induced acute lung injury by ascorbic acid. Am. J. Physiol. Lung Cell Mol. Physiol. 2012. Vol. 303. L20-32.
- Bark B.P., Grände P.-O. The effect of vitamin C on plasma volume in the early stage of sepsis in the rat. Intensive Care Med. Exp. 2014. Vol. 2. P. 11.
- Dey S., Bishayi B. Killing of S.aureus in murine peritoneal macrophages by Ascorbic acid along with antibiotics Chloramphenicol or Ofloxacin: Correlation with inflammation. Microbial. Pathogenesis. 2018. Vol. 115. P. 239-250.
- Barabutis N., Khangoora V., Marik P.E. et al. Hydrocortisone and Ascorbic Acid synergistically protect and repair lipopolysaccharide-induced pulmonary endothelial barrier dysfunction. Chest. 2017. Vol. 152. P. 954-962.
- Reynolds P.S., Fisher B.J., McCarter J. et al. Interventional vitamin C: A strategy for attenuation of coagulopathy and inflammation in a swine multiple injuries model. J. Trauma Acute Care Surg. 2018. Vol. 85. S. 57-67.
- Qi M.Z., Yao Y., Xie R.L. et al. Intravenous Vitamin C attenuates hemorrhagic shock-related renal injury through the induction of SIRT1 in rats. Biochem. Biophys. Res. Commun. 2018. Vol. 501. P. 358-364.
- Ma L., Fei J., Chen Y. et al. Vitamin C Attenuates Hemorrha-gic Shock-induced Dendritic Cell-specific Intercellular Adhesion Molecule 3-grabbing Nonintegrin Expression in Tubular Epithelial Cells and Renal Injury in Rats. Chin. Med. J. 2016. Vol. 129. P. 1731-1736.
- Zhao B., Fei J., Chen Y. et al. Vitamin C treatment attenuates hemorrhagic shock related multi-organ injuries through the induction of heme oxygenase-1. BMC Complement Altern. Med. 2014. Vol. 14. P. 442.
- Zhao B., Fei J., Chen Y. et al. Pharmacological preconditioning with vitamin C attenuates intestinal injury via the induction of heme oxygenase-1 after hemorrhagic shock in rats. PLoS One. 2014. Vol. 9. e99134.
- Ekman T., Risberg B., Bagge U. Ascorbate reduces gastric bleeding after hemorrhagic shock and retransfusion in rats. Eur. Surg. Res. 1994. Vol. 26. P. 187-193.
- Bhandari B., Kohli S.K., Lal V. Protective role of ascorbic acid in hemorrhage-induced cardiovascular depression. Indian J. Physiol. Pharmacol. 2014. Vol. 58. P. 371-375.
- Minor T., Niessen F., Klauke H., Isselhard W. No evidence for a protective effect of ascorbic acid on free radical generation and liver injury after hemorrhagic shock in rats. Shock. 1996. Vol. 5. P. 280-283.
- Daughters K., Waxman K., Gassel A., Zommer S. Anti-oxidant treatment for shock: vitamin E but not vitamin C improves survival. Am. Surg. 1996. Vol. 62. P. 789-792.
- Kremer T., Harenberg P., Hernekamp F. et al. High-dose vitamin C treatment reduces capillary leakage after burn plasma transfer in rats. J. Burn. Care Res. 2010. Vol. 31. P. 470-479.
- Matsuda T., Tanaka H., Williams S. et al. Reduced fluid volume requirement for resuscitation of thirddegree burns with high-dose vitamin C. J. Burn Care Rehabil. 1991. Vol. 12. P. 525-532.
- Dubick M.A., Williams C., Elgjo G.I., Kramer G.C. High-dose vitamin C infusion reduces fluid requirements in the resuscitation of burn-injured sheep. Shock. 2005. Vol. 24. P. 139-144.
- Mohammed B.M., Fisher B.J., Kraskauskas D. et al. Vitamin C promotes wound healing through novel pleiotropic mechanisms. Int. Wound J. 2016. Vol. 13. P. 572-584.
- Spoelstra-de Man A.M.E., Elbers P.W.G., Oudemans-van Straaten H.M. Making sense of early high-dose intravenous vitamin C in ischemia/reperfusion injury. Critical Care. 2018. Vol. 22. P. 70.
- Hao J., Li W.W., Du H. et al. Role of vitamin C in cardioprotection of ischemia/reperfusion injury by activation of mitochondrial KATP channel. Chem. Pharm. 2016. Bull 64. P. 548-557.
- Okazaki T., Otani H., Shimazu T. et al. Ascorbic acid and N-acetyl cysteine prevent uncoupling of nitric oxide synthase and increase tolerance to ischemia/reperfusion injury in diabetic rat heart. Free Radic. Res. 2011. Vol. 45. P. 1173-1183.
- Klein H.H., Pich S., Lindert S. et al. Combined treatment with vitamins E and C in experimental myocardial infarction in pigs. Am. Heart J. 1989. Vol. 118. P. 667-673.
- Doppelfeld I.S., Parnham M.J. Experimental conditions determine effects of ascorbic acid on reperfusion injury: comparison of tissue damage with hemodynamic parameters in rat isolated hearts. Methods Find Exp. Clin. Pharmacol. 1992. Vol. 14. P. 419-430.
- Mickle D.A., Li R.K., Weisel R.D. et al. Myocardial salvage with trolox and ascorbic acid for an acute evolving infarction. Ann. Thorac. Surg. 1989. Vol. 47. P. 553-557.
- Chatziathanasiou G.N., Nikas D.N., Katsouras C.S. et al. Combined intravenous treatment with ascorbic acid and desferrioxamine to reduce myocardial reperfusion injury in an experimental model resembling the clinical setting of primary PCI. Hell. J. Cardiol. 2012. Vol. 53. P. 195-204.
- Skyschally A., Schulz R., Gres P., Korth H.G., Heusch G. Attenuation of ischemic preconditioning in pigs by scavenging of free oxyradicals with ascorbic acid. Am. J. Physiol. Heart Circ. Physiol. 2003. Vol. 284. H698-703.
- Nikas D.N., Chatziathanasiou G., Kotsia A. et al. Effect of intravenous administration of antioxidants alone and in combination on myocardial reperfusion injury in an experimental pig model. Curr. Ther. Res. Clin. Exp. 2008. Vol. 69. P. 423-439.
- Okazaki T., Otani H., Shimazu T. et al. Ascorbic acid and N-acetyl cysteine prevent uncoupling of nitric oxide synthase and increase tolerance to ischemia/reperfusion injury in diabetic rat heart. Free Radic. Res. 2011. Vol. 45. P. 1173-1183.
- Guaiquil V.H., Golde D.W., Beckles D.L., Vitamin C inhibits hypoxia-induced damage and apoptotic signaling pathways in cardiomyocytes and ischemic hearts. Free Radic. Biol. Med. 2004. Vol. 37. P. 1419-1429.
- Gao F., Yao C. L., Gao E. et al. Enhancement of glutathione cardioprotection by ascorbic acid in myocardial reperfusion injury. J. Pharmacol. Exp. Ther. 2002. Vol. 301. P. 543-550.
- Tan D.X., Manchester L.C., Reiter R.J. et al. Ischemia/reperfusion-induced arrhythmias in the isolated rat heart: prevention by melatonin. J. Pineal. Res. 1998. Vol. 25. P. 184-191.
- Karahaliou A., Katsouras C., Koulouras V. et al. Ventricular arrhythmias and antioxidative medication: experimental study. Hell. J. Cardiol. 2008. Vol. 49. P. 320-328.
- Molyneux C.A., Glyn M.C., Ward B.J. Oxidative stress and cardiac microvascular structure in ischemia and reperfusion: the protective effect of antioxidant vitamins. Microvasc. Res. 2002. Vol. 64. P. 265-277.
- Tsai M.S., Huang C.H., Tsai C.Y. et al. Ascorbic acid mitigates the myocardial injury after cardiac arrest and electrical shock. Intensive Care Med. 2011. Vol. 37. P. 2033-2040.
- Tsai M.S., Huang C.H., Tsai C.Y. et al. Combination of intravenous ascorbic acid administration and hypothermia after resuscitation improves myocardial function and survival in a ventricular fibrillation cardiac arrest model in the rat. Acad. Emerg. Med. 2014. Vol. 21. P. 257-265.

/150.jpg)