The article was published on p. 6-11
Introduction
Bronchial asthma has reached epidemic proportions throughout the world. WHO estimated that 235 million people currently suffer from asthma. Asthma is the most common noncommunicable disease among children. The main risk factors for developing asthma are a combination of genetic predisposition with environmental exposure. Nowadays we can observe great improvements in childhood environment, care of children, diagnostic and treatment measures that led to decreasing of the total childhood mortality. However, the prevalence of asthma and allergy in children has increased. Prevalence figures vary in different countries, but up to 20 % of children report recurrent wheezing. In some, wheezing is a childhood-limited problem, but others have persistent life-long asthma [2].
An understanding of the cellular and cytokine interaction during allergic inflammation, especially leading to Th2 — dominated immune response is necessary for predicting the risk of asthma developing, identification of new prophylactic and treatment goals.
Differentiation of effector T cell requires the multiple environmental factors and simultaneous cytokine signals interaction, that provides timulation of the T cell receptor (TCR) and as a consequence significant morphological and functional changes of naive T cells [8].
There are at least six distinct effect or lineages with the irunique features — Th1, Th2, Th9, Th17, Th22 and CD4+CD25+ regulatory T cells (Treg) lineages. Treg cells can come from two sources. Natural Treg (nTreg) cellsare generated during thymicontogeny, where as induced Treg (iTreg) cells differentiated from naïve peripheral T cells when they are activated in the presence of high concentration of trans forming growth factor-β (TGF-β) and interleukin-2 (IL-2). The balance between effector Th cells and Treg cells can critically in fluence the outcomes of many human diseases. Increasing of the level of IL-6 and TGF-β in the microenvironment provokes Th17 cells formation. Activation of T cells by IFN-γ and IL-12 leads to development of Th1 effector cells, presence of IL-4, IL-25, IL-33 and thymic stromal lymphopoietin (TSLP) results in differentiation into Th2 cells [5, 8].
The functions of different types of cells determined by the cytokines, that they produce during antigen stimu–lation. In pathogenesis of allergic inflammation secretion of IL-4, IL-5, IL-6, IL-9, IL-10, IL-31 and IL-13 by Th2 cells leads to allergen-specific synthesis of immunoglobulin E (IgE), favoring the differentiation of eosinophils and activation of development of mast cells [5].
The mammalian target of rapamycin (mTOR, also known as FRAP, RAFT, or RAPT) is an evolutiona–rily conserved serine/threonine kinase that regulates cell metabolism, growth and proliferation according to environmental factors — energy or nutrient avai–lable, cytokine signaling [11]. The mechanism of nutrient sensing function of mTOR is unclear, however it has been suggested, that mTOR may be regulated by intracellular amino –acids, or their metabolites, or by amino acid-activated second messengers. Reduction of cellular ATP levels with the glycolytic inhibitor 2-deoxyglucose inhibits mTOR-dependent process of phosphorylation [4, 10, 11]. Based on significant functions of mTOR in cell development and survival, it was hypothesized that mTOR in naïve T cells may determine antigen recognition outcome and T cells differentiation.
Genetic and biochemical analysis of mTOR revealed that it is a 289 kDa protein that, functions in two structurally and functionally distinct multiprotein complexes termed mTOR complex 1 (mTORC1) and mTOR complex 2 (mTORC2) [14]. mTORC1 contains next components: mTOR, which is the catalytic subunit of the complex; regulatory-associated protein of mTOR (Raptor); mammalian lethal with Sec13 protein 8 (mLST8, also known as GβL); proline-rich AKT substrate 40 kDa (PRAS40); and DEP-domain-containing mTOR-interacting protein (Deptor). mTORC2 consists of six different proteins: mTOR; rapamycin-insensitive companion of mTOR (Rictor); mammalian stress-activated protein kinase interacting protein (mSIN1); protein observed with Rictor-1 (Protor-1); mLST8; and Deptor. The functions of each component still are unclear, but there is some evidence that Rictor and mSIN1 stabilize each other, establishing the structural organization of mTORC2 [14].
In a generalscheme, activation of mTORC1 is achieved through the phosphatidylinositide-3-kinase (PI3K), phosphoinositide-dependent kinase-1 (PDK1), and proteinkinase B (PKB alsoknown as AKT) signa–ling. Growth factor or cytokine signaling leads to the activation of PI-3K, which activates the PDK1, which then promotes the phosphorylation of Akt. The proximal activation motif phosphorylation of Akt leads to the phosphorylation and inactivation of tuberous sclerosis complex (TSC). TSC — a heterodimer consisting of hamartin (Tuberous sclerosis 1 — TSC1) and tuberin (Tuberous sclerosis 2 — TSC2), functions as aninhibitor of mTOR by inactivating Ras homolog, expressed in brain (Rheb) [3, 9]. Besides this, formation of mTORC1 requires activation of regulator complex and the four Ras-like GTPases (RAGs A-D), that detect the level and availability of essential amino acids. In the case of TCR stimulation and all necessary factors are available, activation of mTOR occurs, that leads to inhibition of Forkhead box P3 (FOXP3) expression and differentiation of effector T cell. Otherwise, mTOR inhibition provokes FOXP3 expression and development of FOXP3 + regulatory T cells [7].
Function of mTORC1 is regulation of cell growth (accumulation of cell mass) and proliferation through coordination of protein anabolism, nucleotide biosynthesis, lipogenesis, biosynthesis of organelles, glycolysis and by limiting catabolic processes such as autophagy [1, 14]. The mechanism how mTORC1 promotes protein synthesis is by phosphorylating the initiation factor 4E (eIF4E)-binding protein 1 (4E-BP1) and the p70 ribosomal S6 kinase 1 (S6K1), positive and negative regulators of protein synthesis. Phosphorylation of S6K1 promotes activation of the downstream 40S ribosomal protein S6 and increases translation of mRNA. The phosphorylation of 4E-BP1 prevents its binding to eIF4E, enabling eIF4E to promote translation [1, 19]. mTOR controls mitochondrial oxidative function, oxygen consumption and cellular ATP levels. Function of mTORC2 is regulation of cytoskeleton organization by controlling of actin polymerization. In turn, activation of mTORC2 inhibits two transcription factors — Forkhead Factor 1 (FOXO1) and Kruppell-like factor 2 (KLP) that maintain the T cell quiescent state [14, 19].
Polymorphism rs11121704 (T > C) in intron have been mostly frequently studied as genetic risk factor for the carcinogenesis and clinical outcomes of cancer patients in USA and China populations [13]. Polymorphism rs11121704 TT genotype was associated with poor clinical outcomes, such as death, metastasis, resistant to chemotherapy, and toxicity (OR = 1.53, р = 0.044).We hypothesized that polymorphism rs11121704 in mTOR gene would be associated with childhood asthma and with allergic phenotype. To test this hypothesis, we exa–mined differences in the frequency of this SNP between children without allergic pathology (control group) and children with eczema plus asthma.
Material and Methods
The research includes 91 children 5 to 18 years of age from subspecialty department at Kiev Children’s Hospital and 87 control subjects from the general population. All children had symptoms of asthma, eczema or symptoms of eczema in anamnesis, allergic rhinitis, or at least one positive skin prick test result to a panel of 15 aeroallergens. Skin prick tests were carried out and interpreted by Global Allergy and Asthma European Network protocol, developed using a common panel of inhalant allergens based on published practice guidelines, the European Academy of Allergy and Clinical Immunology (EAACI) Position Paper, the Nordic standards and the International Study of Asthma and Allergies in Childhood (ISAAC) phase II protocol. Parents signed an informed consent prior to their inclusion in the study.
Outcome measures
The definition of eczema was adapted from a valida–ted questionnaire (ISAAC) and included a parental report of the child’s scratching and redness «raised bumps» or dry skin/scaling during the childhood. Cases with eczema included children with a physician’s diagnosis or a parental report of eczema. Physical examination fin–dings considered to be consistent with eczema included erythema, papulation, excoriations, and/or lichenification. Testing for asthma were performed; the definition included a parental report of persistent wheezing (≥ 2 wheezing episodes not associated with a cold or upper respiratory tract infection), spirometry (a reduced FEV1 and FEV1/FVC ratio, positive test with β-2 agonists), increased IgE level, positive skin prick tests with aeroallergens.
Selection of SNP
An rs11121704 in mTOR genewas selected for genotyping, as it hypothetically can affect Th2 differentiation and is reported to be common in European populations.
DNA extraction
Buccal epithelium was taken using buccal brushes with the following freezing of samples and their storage at –20 °C. DNA for genotyping was extracted from the samples using Isogene kits (Russian Federation) accor–ding to manufacturer’s protocol. The concentration of total DNA was determined using a NanoDrop spectrophotometer ND1000 (NanoDrop Technologies Inc., USA).
qPCR Genotyping
Amplification reactions were performed using a 7500 Fast Real-time PCR System (Applied Biosystems, USA) in a final reaction volume of 20 μl, which contained 2X TaqMan Universal Master Mix (Applied Biosystems, USA), assay C_31720978_30and the template cDNA. The thermal cycling conditions involved a denaturation step at 95 °C for 20 s, followed by 40 cycles of amplification at 95 °C for 3 s and 60 °C for 30 s. Analysis of the data was carried out with 7500 Fast Real-Time PCR Software.
Statistical analysis
χ2 test was performed to investigate if there was any difference in the frequency of the genotype and allele between the patient group with allergic diseases and the normal control group. A p-value of less than 0.05 (p < 0.05) was considered statistically significant. Frequency distribution of genotypes was analyzed using Pearson chi-square test with Excel 2000 software. SNP Analyzer (web-based software) was used to examine Hardy-Weinberg equilibrium.
Results
Clinic
100 % of children have atopic dermatitis in history, 38.5 % of children have manifestations of atopic dermatitis in the form of rashes, dry skin, peeling, pruritus, lichenification during last year. 72 (73.4 %) of children were in a state of exacerbation of asthma, manifested in wheezing and cough (of which 40 (55.5 %) had mild exa–cerbation severity, 31 (43.1 %) had moderate seve–rity of exacerbation, 1 (1.4 %) had severe exacerbations), 28 % of children had remission. 60 (83.3 %) of patients had decreased FEV1 (< 80 %), 34 (47.2 %) had positive test with β-2 agonist. Other concomitant allergic patho–logy was determined in 46 (46.9 %) children. 23 (25.3 %) children had allergic rhinitis, 18 (18.4 %) had persistent allergic rhinitis, 5 (5.5 %) — seasonal. Also 4 (4.4 %) children had acute allergic urticaria in anamnesis data, 9 (9.9 %) children — food allergy.
Genotyping
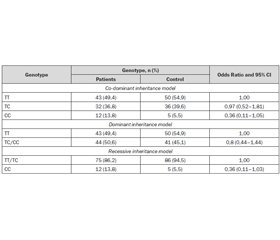
We found that SNP was in Hardy-Weinberg equilibrium. Our main results are as follows: 50 (54.9 %) of patients and 43 (49.4 %) of control group had major allele of rs11121704 in mTOR (TT), 36 (39.6 %) of patients and 32 (36.8 %) of control group, respectively, had he–terozygous allele (TC), and 5 (5.5 %) and 12 (13.8 %) had minor allele (CC). Variants of rs11121704 CC genotype in the mTORgene were 2,5 times more likely in the control group than among patients (table 1).
Statistical analysis was conducted to identify possible associations of genetic markers with the presence of concomitant allergic diseases. The research group consisted of 22 patients with other allergic diseases, and control group — 69. Using the chi-square test with two degrees of freedom we found statistically significant differences in the distribution of genotypes in patients with concomitant allergic diseases and groups of patients without it (chisquare = 10.578; statistical significance = 0.005) (table 2).
Discussion
Evidence from the literature has established that mTOR function is necessary for Th1 and Th2 effector T cell differentiation and plays a critical role in eczema and possibly in the phenotype of eczema plus asthma.
Delgoffe and colleagues suggest opposite regulation of mTORC1 and mTORC2 on T cell differentiation into Th1, Th2, and Th17. They found that mTOR deficient CD4+ T cells were unable to upregulate subset specific transcription factors, such as GATA-3, T-bet, and RORgt. When Rheb, a positive upstream regulator of mTORC1, was deleted in T cells, these cells failed to generate Th1, and Th17 responses, but retained their ability to differentiate to Th2. Furthermore, Rheb (–/–) T cells responded to cytokine IL-4 release by activating the STAT 6 signaling, that prove their ability to differentiate into Th2 cells. Otherwise, when mTORC2 signa–ling was deleted in T cells, they failed to generate TH2 cells in vitro and in vivo but retain their ability to generate TH1 or TH17 response. Also, it has been shown, thatmTORC1 promotes STAT4 and STAT3 activation media–ted by –IL-12 and IL-6, respectively, while IL-4 mediated STAT6 activation requires mTORC2 activation [8, 9].
Another investigation showed that conditional deletion of rictor (an essential subunit of mTORC2) impaired differentiation into Th1 and Th2 cells without diversion into FoxP3+ status or substantial effect on Th17 cell differentiation [15].
Salmond et al. described the role for mTOR signa–ling in IL-330-dependent Th2 and innate lymphoid cell (ILC) effector responses. They found that IL-33 directly activated mTOR in a PI3K p110δ-dependent manner. It was shown that IL-33 directly expands the population of lung ILC, which mediates airway inflammation. Furthermore, inhibition of mTOR reduced IL-33-driven IL-5 and IL-13 expression by both TH2 cells and ILCs in vitro. IL-33-induced airway inflammation was mediated by ILCs and that rapamycin reduced ILC accumulation, macrophage and eosinophil infiltration, cytokine secretion, and mucus deposition in the lung [20].
Interesting, that mTOR is also involved in the process of growth, maturation of mast cells and cytokine secretion. Shin et al, in their investigation found that TSC1 is a critical regulator for mTOR signaling in mast cells downstream of FcεRI and c-Kit, and differentially controls mast cell degranulation and cytokine production. TSC1-deficiency results in failed mast cell degranulation, but enhanced cytokine production in vitro and in vivo after FcεRI engagement. Furthermore, TSC1 is critical for mast cell survival through multiple pathways of apoptosis including the down-regulation of p53, miR-34a, reactive oxygen species, and the up-regulation of Bcl-2 [21].
These data demonstrate the significant role of mTOR in regulation of cytokine signals, differentiation and development of effector Th cells, maturation and degra–nulation of mast cells, regulation of the process of antigen presentation via autophagy, that subsequently lead to formation of Th2 cell polarized effector phenotype. Consistent with this, the ability of rapamycin-derivatives to inhibit asthma has been investigated in experimental animal model, using a clinically relevant house dust mite (HDM)-induced and ovalbumin (OVA)-induced model of murine disease.
Fredrikkson et al described in his published study, that inhibition of mTOR signaling by rapamycin has pa–radoxical effects on the manifestations of HDM-induced asthma. Inhibition of mTOR signaling with rapamycin prior to the induction of asthma effectively suppressed airway inflammation, goblet cell hyperplasia and airway hyperreactivity, whereas inhibition of mTOR signaling in established asthma exacerbated airway inflammation and airway hyperreactivity, but did not modify HDM-induced increases in goblet cell hyperplasia. Rapamycin also inhibits TGF-a-induced pulmonary fibrotic responses, which could contribute to sub-epithelial fibrosis and airway remodeling. Similarly, mTOR signaling may modulate angiogenesis and lymphangiogenesis, both of which play important roles in asthma pathogenesis. Inhibition of mTOR blocks the synthesis of vascular endothelial growth factor (VEGF), an angiogenesis factor that induces an asthma phenotype in mice [12]. Mushaben E.M. et al. showed that inhibition of mTOR by rapamycin prevented HDM-induced allergic asthma in mice. Also rapamycin suppressed IgE and IL-4 level was augmented in mice that were sensitized to HDM [16].
It also has been reported that rapamycin blocks OVA-induced increases in bronchial inflammation, airway hyperresponsiveness, levels of Th2 and proinflammatory cytokines and adhesion molecules, and vascular permeability, suggesting that rapamycin can inhibit main symptoms of allergic asthma [6].
In this article, we determined the frequency of the rs11121704 SNP and showed that minor allele occurs 2.5 times more likely in the control group than among patients, although there was no statistical significance. Using the chi-square test with two degrees of freedom we found statistically significant differences in the distribution of genotypes in patients with concomitant allergic diseases and groups of patients without it (chi-square = = 10.578; statistical significance = 0.005).
Conclusions
It is widely accepted that mTOR plays a critical role in growth, differentiation, survival and cell metabolism. More recent works have demonstrated that mTOR may act as a susceptibility factor to generate aberrant Th2 allergic responses to antigen. Due to its function, mTOR pathway is attractive target for the prevention and treatment of allergic disorders. Moreover, positive results of rapamycin administration for prevention and treatment of asthma were already obtained. Although progress in understanding of mTOR functions has been made, but much is still unknown regarding differential regulation of mTOR complexes, ways of influence on this process. Further researches are required to precisely delineate the role of mTOR in allergic inflammatory response.
Variants of rs11121704 genotype CC in the gene mTOR were 2.5 times more likely in the control group than among patients. Our research has demonstrated protective significance of CC genotype of mTOR polymorphism, thus suggesting that this polymorphism may have a predictive value in the development of allergic diseases affecting the expression of that gene. Also we found statistically significant differences in the distribution of genotypes in patients with concomitant allergic diseases and groups of patients without it.
There by, polymorphisms in mTOR may in depen–dently serve as perspective marker to prognostication the development of asthma and other allergic diseases among children.
Список литературы
1. Averous J., Proud C.G. When translation meets transformation: the mTOR story [Text] / J. Averous, C.G. Proud // Oncogene. — 2006. — 25. — P. 6423-6435.
2. Belsky D.W., Sears M.R. The potential to predict the course of childhood asthma [Text] / D.W. Belsky, M.R. Sears // Expert Rev. Respir. Med. — 2014 Apr. — 8(2). — P. 137-41.
3. Betz C., Hall M.N. Where is mTOR and what is it doing there? [Text] / C. Betz, M.N. Hall // J. Cell. Biol. — 2013. — 203(4). — P. 563-574.
4. Beugnet A., Tee A.R., Taylor P.M., Proud C.G. Regulation of targets of mTOR (mammalian target of rapamycin) signalling by intracellular amino acid availability [Text] / A. Beugnet, A.R. Tee, P.M. Taylor, C.G. Proud // Biochem. J. — 2003. — 372. — P. 555-566.
5. Brand E.B., Sivaprasad U. Th2 Cytokines and Atopic Dermatitis [Text] / E.B. Brand, U. Sivaprasad // J. Clin. Cell. Immunol. — 2011. — 2(3). — P. 110.
6. O’Brien T.F., Xiao-Ping Zhong. The Role and Regulation of mTOR in T Lymphocyte Function [Text] / T.F. O’Brien, Xiao-Ping Zhong // Arch. Immunol. Ther. Exp. (Warsz). — 2012. — 60(3). — P. 173-181.
7. Choi Y.H., Jin G.Y., Li L.C., Yan G.H. Inhibition of protein kinase C delta attenuates allergic airway inflammation through suppression of PI3K/Akt/mTOR/HIF-1 alpha/VEGF pathway [Text] / Y.H. Сhoi, G.Y. Jin, L.C. Li, G.H. Yan // PLoS One. — 2013. — 29. — 8(11). — e81773. — doi: 10.1371.
8. Cobbold S.P. ThemTOR pathway and integrating immune re–gulation [Text] / S.P. Cobbold // Immunology. — 2013. — 140(4). — P. 391-8.
9. Delgoffe G.M., Kole T.P., Zheng Y., Zarek P.E., Matthews K.L., Xiao B., Worley P.F., Kozma S.C., Powell J.D. mTOR differentially regulates effector and regulatory T cell lineage commitment [Text] / G.M. Delgoffe, T.P. Kole, Y. Zheng, P.E. Zarek, K.L. Matthews, B. Xiao, P.F. Worley, S.C. Kozma, J.D. Powell // Immunity. — 2009. — 30(6). — P. 832844. — doi: 10.1016/j.immuni.2009.04.014.
10. Delgoffe G.M., Pollizzi K.M., Waickman A.D., Heikamp E., Meyers D.J., Horton M.R., Bo Xiao, Worley P.F., Powell J.D. The mammalian Target of Rapamycin (mTOR) regulates T helper cell differentiation through the selective activation of mTORC1 and mTORC2 signaling [Text] / G.M. Delgoffe, K.M. Pollizzi, A.D. Waickman, E. Heikamp, D.J. Meyers, M.R. Horton, Bo Xiao, P.F. Worley, J.D. Powell // Nature immunology. — 2011. — 12(4). — P. 295-303.
11. Dennis P.B., Jaeschke A., Saitoh M., Fowler B., Kozma S.C., Thomas G. Mammalian TOR: a homeostatic ATP sensor [Text] / P.B. Dennis, A. Jaeschke, M. Saitoh, B. Fowler, S.C. Kozma, G. Thomas // Science. — 2001. — 294. — P. 1102-5.
12. Fingar D.C., Blenis J. Target of rapamycin (TOR): an integrator of nutrient and growth factor signals and coordinator of cell growth and cell cycle progression [Text] / D.C. Fingar, J. Blenis // Oncogene. — 2004. — 23. — P. 3151-3171.
13. Fredriksson K., Fielhaber J.A., Lam J.K., Yao X., Meyer K.S. et al. Paradoxical Effects of Rapamycin on Experimental House Dust Mite-Induced Asthma [Text] / K. Fredriksson, J.A. Fielhaber, J.K. Lam, X. Yao, K.S. Meyer et al. // PLoS ONE. — 2012. — 7(5). — e33984. — doi:10.1371/journal.pone.0033984.
14. Huang L., Huang J., Wu P., Li Q., Rong L. et al. Association of genetic variations in mTOR with risk of childhood acute lymphoblastic leukemia in a Chinese population [Text] / L. Huang, J. Huang, P. Wu, Q. Li, L. Rong et al. // Leuk Lymphoma. — 2012. — 53. — 947-951. — doi: 10.3109/10428194.2011.628062.
15. Laplante M., Sabatini D.M. mTOR signaling at a glance [Text] / M. Laplante, D.M. Sabatini // J. Cell. Sci. — 2009. — 122. — P. 3589-3594. — doi: 10.1242/jcs.051011.
16. Lee K., Gudapati P., Dragovic S., Spencer C., Joyce S., Killeen N., Magnuson M.A., Boothby M. Mammalian target of rapamycin protein complex 2 regulates differentiation of Th1 and Th2 cell subsets via distinct signaling pathways [Text] / K. Lee, P. Gudapati, S. Dragovic, C. Spencer, S. Joyce, N. Killeen, M.A. Magnuson, M. Boothby // Immunity. — 2010. — 32(6). — P. 743-753.
17. Mushaben E.M., Brandt E.B., Khurana Hershey G.K., Le Cras T.D. Differential effect of rapamycin and dexamethasone in mouse models of established allergic athma [Text] / E.M. Mushaben, E.B. Brandt, G.K. Khurana Hershey, T.D. Le Cras // PLo ONE. — 2013. — 8(1). — e54426. — doi:10.1371/journal.pone.0054426.
18. Nakano H., Ushio H. An unexpected role for autophagy in degranulation of mast cells / H. Nakano, H. Ushio // Autophagy. — 2011. — 7. — 6. — P. 657-659.
19. Poon A.H., Chouiali F., Tse S.M., Litonjua A.A., Hussain S.N., Baglole C.J., Eidelman D.H., Olivenstein R., Martin J.G., Weiss S.T., Hamid Q., Laprise C. Genetic and histologic evidence for autophagy in asthma pathogenesis [Text] / Poon A.H., F. Chouiali, S.M. Tse, A.A. Litonjua, S.N. Hussain, C.J. Baglole, D.H. Eidelman, R. Olivenstein, J.G. Martin, S.T. Weiss, Q. Hamid, C. Laprise // J. Allergy. Clin. Immunol. — 2012. — 129(2). — P. 569-71. — doi: 10.1016/j.jaci.2011.09.035.
20. Soliman G.A. The role of mechanistic target of rapamycin (mTOR) complexes signaling in the immune responses [Text] / G.A. Soliman // Nutrients. — 2013. — 5(6). — P. 2231-57.
21. Salmond J., Mirchandani A.S., Besnard A.G., Bain C.C., Thomson N.C., Liewa F.E. IL-33 induces innate lymphoid cell-mediated airway inflammation by activating mammalian target of rapamycin[Text] / J. Salmond, A.S Mirchandani., A.G. Besnard, C.C. Bain, N.C. Thomson, F.E. Liewa // J. Allergy Clin. Immunol. — 2012. — 130(5). — P. 1159-1166.
22. Shin J., Pan H., Xiao-Ping Zhong. Regulation of mast cell survival and function by tuberous sclerosis complex 1 [Text] / J. Shin, H. Pan, Xiao-Ping Zhong // Blood. — 2012. — 119(14). — P. 3306-3314.

/9.jpg)